Chapter 8: An Introduction to Metabolism
- characteristic of all living things
- controls the material and energy resources within a cell
-Metabolic pathway: describes the steps in chemical reactions beginning with the starting molecule and through a series of reactions that involve specific enzymes to catalyze each step, and ending with a product molecule.
- metabolism is regulated through these enzymes
-Catabolism: metabolic processes that involve the degradation of complex molecules into simpler molecules, and result in the release of energy. (tip to remember catabolism: catastrophes cause damage, catabolic pathways break down molecules)
-Cellular respiration is a catabolic pathway: glucose or other energy storing molecules are broken down to release their energy for cellular work
-Anabolism (Biosynthetic Pathways): metabolic processes that involve the building of complex molecules from simpler molecules, and require an input of energy. (tip to remember anabolism: anabolic steroids cause larger muscles to grow, anabolic pathways result in larger molecules being built)
- The formation of amino acids from simple molecules and then the formation of proteins from amino acids are examples of an anabolic reaction.
- The same is true for any reaction that involves building a monomer, or building a polymer from those monomers.
-Catabolic pathways provide the energy needed by the anabolic pathways.
-Bioenergetics: the study of how energy flows through living organisms.
- 8.1: An organism's metabolism transforms matter and energy, subject to the laws of thermodynamics
- characteristic of all living things
- controls the material and energy resources within a cell
-Metabolic pathway: describes the steps in chemical reactions beginning with the starting molecule and through a series of reactions that involve specific enzymes to catalyze each step, and ending with a product molecule.
- metabolism is regulated through these enzymes
-Catabolism: metabolic processes that involve the degradation of complex molecules into simpler molecules, and result in the release of energy. (tip to remember catabolism: catastrophes cause damage, catabolic pathways break down molecules)
-Cellular respiration is a catabolic pathway: glucose or other energy storing molecules are broken down to release their energy for cellular work
-Anabolism (Biosynthetic Pathways): metabolic processes that involve the building of complex molecules from simpler molecules, and require an input of energy. (tip to remember anabolism: anabolic steroids cause larger muscles to grow, anabolic pathways result in larger molecules being built)
- The formation of amino acids from simple molecules and then the formation of proteins from amino acids are examples of an anabolic reaction.
- The same is true for any reaction that involves building a monomer, or building a polymer from those monomers.
-Catabolic pathways provide the energy needed by the anabolic pathways.
-Bioenergetics: the study of how energy flows through living organisms.
Forms of Energy
Energy: the capacity to cause change
- Life depends on an organism's capability to transform energy
- Used to move matter against opposing forces like gravity and friction
-Kinetic energy: energy that results from movement, movement performs work by transferring motion to another object
-Thermal energy: kinetic energy that refers specifically to the random movement of atoms or molecules
- heat: the transfer of thermal energy to another object
-Solar energy: light is transformed into chemical energy during the process of photosynthesis.
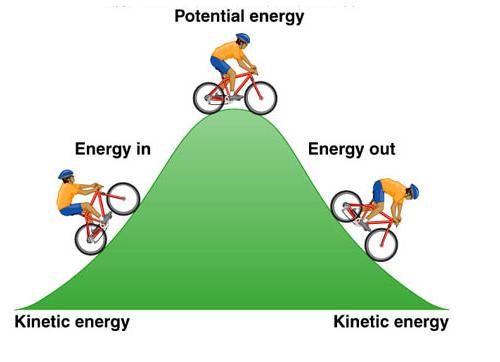
-Potential energy: energy than an object possesses because of its location or structure (not kinetic!)
-Chemical energy: molecules contain potential energy stored within their bonds that can be released
during chemical reactions that break these bonds.
-During cellular respiration oxygen is used to breakdown food molecules, resulting in chemical energy
that can be utilized by the cell
-see how the ATP produced by cellular respiration can be utilized for energy that drives anabolic pathways
with the movie below:
Energy: the capacity to cause change
- Life depends on an organism's capability to transform energy
- Used to move matter against opposing forces like gravity and friction
-Kinetic energy: energy that results from movement, movement performs work by transferring motion to another object
-Thermal energy: kinetic energy that refers specifically to the random movement of atoms or molecules
- heat: the transfer of thermal energy to another object
-Solar energy: light is transformed into chemical energy during the process of photosynthesis.
-Potential energy: energy than an object possesses because of its location or structure (not kinetic!)
-Chemical energy: molecules contain potential energy stored within their bonds that can be released
during chemical reactions that break these bonds.
-During cellular respiration oxygen is used to breakdown food molecules, resulting in chemical energy
that can be utilized by the cell
-see how the ATP produced by cellular respiration can be utilized for energy that drives anabolic pathways
with the movie below:
The Laws of Energy Transformation
Thermodynamics: the study of how energy is transformed
-isolated system: the system cannot exchange energy or matter with its surroundings (no inputs or outputs!)
-open system: energy and matter can be exchanged between the system and the surroundings (both inputs and outputs!)
-organisms are open systems
The first law of thermodynamics (Principle of conservation of energy): energy can be transferred and transformed, but it cannot be created nor destroyed
- energy cannot be recycled within an organism because it is lost to the surroundings as heat, so only some is
available to "do work"
The second law of thermodynamics: every energy transfer or transformation increases the entropy of the universe.
-entropy: a measure of disorder or randomness
-the universe favors disorder (higher entropy), though not highly obvious when considering heat loss and other less ordered forms of matter
- processes that do not require energy input (ie: catabolism) are increasing disorder (just like a catastrophic tornado increases disorder), and therefore favored universally.
-Spontaneous processes: processes that occur with no energy input, increased entropy. (energetically favorable)
-Nonspontaneous processes: processes that require energy input, decreased entropy. (energetically unfavorable)
Thermodynamics: the study of how energy is transformed
-isolated system: the system cannot exchange energy or matter with its surroundings (no inputs or outputs!)
-open system: energy and matter can be exchanged between the system and the surroundings (both inputs and outputs!)
-organisms are open systems
The first law of thermodynamics (Principle of conservation of energy): energy can be transferred and transformed, but it cannot be created nor destroyed
- energy cannot be recycled within an organism because it is lost to the surroundings as heat, so only some is
available to "do work"
The second law of thermodynamics: every energy transfer or transformation increases the entropy of the universe.
-entropy: a measure of disorder or randomness
-the universe favors disorder (higher entropy), though not highly obvious when considering heat loss and other less ordered forms of matter
- processes that do not require energy input (ie: catabolism) are increasing disorder (just like a catastrophic tornado increases disorder), and therefore favored universally.
-Spontaneous processes: processes that occur with no energy input, increased entropy. (energetically favorable)
-Nonspontaneous processes: processes that require energy input, decreased entropy. (energetically unfavorable)
Biological Order and Disorder:
-living systems increase the entropy of their surroundings
- though cells build monomers and then use monomers to build highly complex and organized structural polymers, they also take in more organized forms of matter and energy from their surroundings and replace them with less ordered forms- like taking in food and breaking it down or transforming energy into heat that enters the surroundings.
-Although we have evolved to become more complex and organized organisms, with lower entropy, it doesn't violate the second law when we consider that we are talking about the universe and not just organisms. As long as the universe continues to increase in entropy, the law is being upheld.
Gibbs free energy of a system= G
Free Energy: energy within a system that can perform work (where temperature and pressure are uniform, which is the normal in a living cell)
-living systems increase the entropy of their surroundings
- though cells build monomers and then use monomers to build highly complex and organized structural polymers, they also take in more organized forms of matter and energy from their surroundings and replace them with less ordered forms- like taking in food and breaking it down or transforming energy into heat that enters the surroundings.
-Although we have evolved to become more complex and organized organisms, with lower entropy, it doesn't violate the second law when we consider that we are talking about the universe and not just organisms. As long as the universe continues to increase in entropy, the law is being upheld.
- 8.2: The free-energy change of a reaction tells us whether or not the reaction occurs spontaneously
Gibbs free energy of a system= G
Free Energy: energy within a system that can perform work (where temperature and pressure are uniform, which is the normal in a living cell)
-Based on your change (∆) in energy (G), you can predict whether or not a reaction will occur spontaneously
-Negative ∆G values = spontaneous (energetically favorable, no E input)
-Positive or zero ∆G values= nonspontaneous (energetically unfavorable, E input required),
Free Energy, Stability, and Equilibrium
∆G(system)= G (final)-G(initial)
-Negative ∆G values mean there was a loss of free energy (reactants have higher G than products)
-Think of this as energy released by the breaking of bonds(water flowing from the dam, less potential energy)
-More stability, less likely to change
-Positive ∆G values mean there was a gain of free energy (products have higher G than reactants)
-Think of this as energy being stored in the bonds formed.(water being held back by a dam, potential energy)
-Less stability, more likely to change to a more stable state
Stability = more disorder (although this seems to contradict itself)
-The universe favors disorder
*Think of a ball being held at the top of a hill: unstable, high potential energy, higher G*
-that ball is likely to roll to the bottom (stable, no potential energy, lower G)
-The ball will not roll back up the hill on its own, you must put in work
*watch the beginning of the video below (maybe on mute because of the music)*
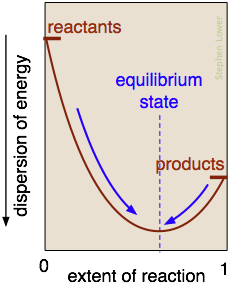
-the blue dye is dropped into the water (droplets are unstable, higher G)
-dye becomes dispersed throughout the water (stable, lower G)
-the dye will not return to droplets because the system is at equilibrium (lowest G, max stability), and therefore,
though seemingly less organized, it is more stable because it will not change anymore.
-Negative ∆G values = spontaneous (energetically favorable, no E input)
-Positive or zero ∆G values= nonspontaneous (energetically unfavorable, E input required),
Free Energy, Stability, and Equilibrium
∆G(system)= G (final)-G(initial)
-Negative ∆G values mean there was a loss of free energy (reactants have higher G than products)
-Think of this as energy released by the breaking of bonds(water flowing from the dam, less potential energy)
-More stability, less likely to change
-Positive ∆G values mean there was a gain of free energy (products have higher G than reactants)
-Think of this as energy being stored in the bonds formed.(water being held back by a dam, potential energy)
-Less stability, more likely to change to a more stable state
Stability = more disorder (although this seems to contradict itself)
-The universe favors disorder
*Think of a ball being held at the top of a hill: unstable, high potential energy, higher G*
-that ball is likely to roll to the bottom (stable, no potential energy, lower G)
-The ball will not roll back up the hill on its own, you must put in work
*watch the beginning of the video below (maybe on mute because of the music)*
-the blue dye is dropped into the water (droplets are unstable, higher G)
-dye becomes dispersed throughout the water (stable, lower G)
-the dye will not return to droplets because the system is at equilibrium (lowest G, max stability), and therefore,
though seemingly less organized, it is more stable because it will not change anymore.
Chemical Equilibrium: when a reaction proceeds forward and backward at the same rate, no net change in the concentration of products and reactants.
-most reactions are reversible (can proceed in the forward direction: reactants--> products, or reverse direction reactants<--products)
-A system is most stable when it is at equilibrium, all systems will proceed in the direction of equilibrium.
-Lowest amount of free energy (G)
-Systems never move away from equilibrium spontaneously
-most reactions are reversible (can proceed in the forward direction: reactants--> products, or reverse direction reactants<--products)
-A system is most stable when it is at equilibrium, all systems will proceed in the direction of equilibrium.
-Lowest amount of free energy (G)
-Systems never move away from equilibrium spontaneously
Free Energy and Metabolism:
Exergonic and Endergonic Reactions in Metabolism:
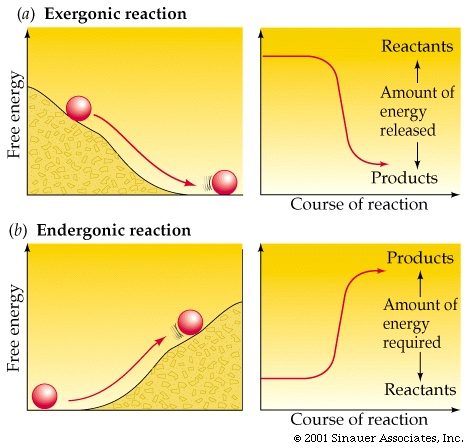
Exergonic reactions: a reaction that results in a net release of free energy (decrease in G, negative ∆G), occur spontaneously
- The magnitude ∆G represents the amount of work the reaction can do. (though some is lost as heat)
- potential energy is released, products are of lower free energy than reactants
Endergonic reactions: a reaction that results in a net gain of free energy (increase in G, negative ∆G), not spontaneous
- The magnitude of ∆G represents the amount of work required for the reaction to occur
- potential energy is stored, products are of higher free energy than reactants.
Exergonic and Endergonic Reactions in Metabolism:
Exergonic reactions: a reaction that results in a net release of free energy (decrease in G, negative ∆G), occur spontaneously
- The magnitude ∆G represents the amount of work the reaction can do. (though some is lost as heat)
- potential energy is released, products are of lower free energy than reactants
Endergonic reactions: a reaction that results in a net gain of free energy (increase in G, negative ∆G), not spontaneous
- The magnitude of ∆G represents the amount of work required for the reaction to occur
- potential energy is stored, products are of higher free energy than reactants.
Equilibrium and Metabolism:
-Metabolism as a whole can never be in equilibrium
-Systems at equilibrium can do no work, so if metabolism's reactions reached equilibrium, an organism would die.
-Preventing Equilibrium in a cell:
- constant transport of materials in and out the cell
-Use of products in the subsequent reaction as a reactant, prevents build up of products
1) Chemical: pushing nonspontaneous reactions to occur
2) Transport: transporting substances against gradients into or out of the cell across the cell membrane
3) Mechanical: movement of cilia/flagella, muscle contraction, chromosomes during cell reproduction
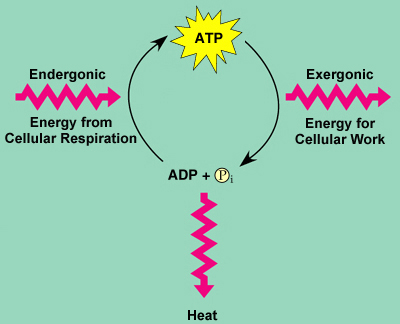
Energy coupling: the use of an exergonic reaction to drive exergonic reaction
- ATP is responsible for coupled reactions by provided initial energy
The Structure and Hydrolysis of ATP:
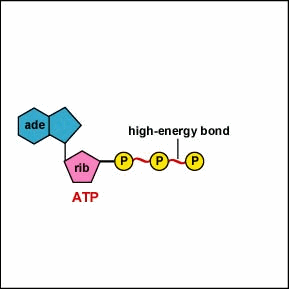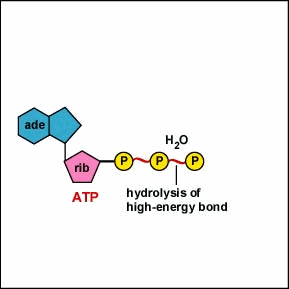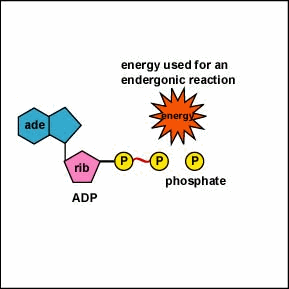
ATP (adenosine triphosphate): molecule composed of ribose, the nitrogenous base adenine, and 3 phosphate groups
-bonds between phosphate groups can be hydrolyzed (broken by the addition of a water molecule)
-The hydrolysis of these bonds result in a release of energy because the products adenosine diphosphate and inorganic phosphate are lower in free energy (more stable) than ATP and water.
-ATP has high free energy because the 3 phosphate groups in the molecule are negatively charged and crowded together.
-the repulsion between the negatively charged phosphate groups makes the ATP molecule relatively unstable.
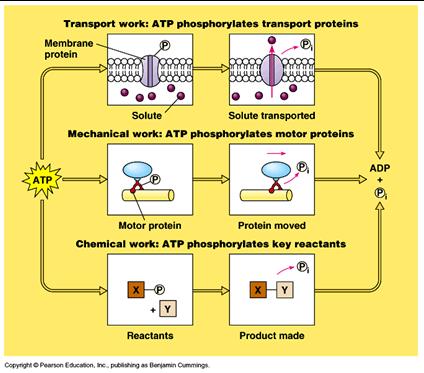
How the hydrolysis of ATP Performs Work:
-proteins in cells harness the energy released by hydrolysis of ATP to perform work
-if the ∆G of an endergonic reaction is less than the amount of energy released by ATP hydrolysis, then the two reactions can be coupled so overall ∆G is still negative.
-this usually involves phosphorylation: the transfer of the phosphate group from ATP to another molecule
-The phosphorylated molecule becomes less stable and capable of doing work
-Transport proteins
-Motor proteins
-Metabolism as a whole can never be in equilibrium
-Systems at equilibrium can do no work, so if metabolism's reactions reached equilibrium, an organism would die.
-Preventing Equilibrium in a cell:
- constant transport of materials in and out the cell
-Use of products in the subsequent reaction as a reactant, prevents build up of products
- 8.3: ATP powers cellular work by coupling exergonic and endergonic reactions:
1) Chemical: pushing nonspontaneous reactions to occur
2) Transport: transporting substances against gradients into or out of the cell across the cell membrane
3) Mechanical: movement of cilia/flagella, muscle contraction, chromosomes during cell reproduction
Energy coupling: the use of an exergonic reaction to drive exergonic reaction
- ATP is responsible for coupled reactions by provided initial energy
The Structure and Hydrolysis of ATP:
ATP (adenosine triphosphate): molecule composed of ribose, the nitrogenous base adenine, and 3 phosphate groups
-bonds between phosphate groups can be hydrolyzed (broken by the addition of a water molecule)
-The hydrolysis of these bonds result in a release of energy because the products adenosine diphosphate and inorganic phosphate are lower in free energy (more stable) than ATP and water.
-ATP has high free energy because the 3 phosphate groups in the molecule are negatively charged and crowded together.
-the repulsion between the negatively charged phosphate groups makes the ATP molecule relatively unstable.
How the hydrolysis of ATP Performs Work:
-proteins in cells harness the energy released by hydrolysis of ATP to perform work
-if the ∆G of an endergonic reaction is less than the amount of energy released by ATP hydrolysis, then the two reactions can be coupled so overall ∆G is still negative.
-this usually involves phosphorylation: the transfer of the phosphate group from ATP to another molecule
-The phosphorylated molecule becomes less stable and capable of doing work
-Transport proteins
-Motor proteins
The Regeneration of ATP:
- ATP is a renewable resource because a phosphate group can be added back to ADP
-this is an energonic reaction that requires energy
-the energy needed comes from exergonic reactions.
- ATP is a renewable resource because a phosphate group can be added back to ADP
-this is an energonic reaction that requires energy
-the energy needed comes from exergonic reactions.
- 8.4: Enzymes speed up metabolic reactions by lowering energy barriers
Enzyme: a macromolecule (protein) that acts as a catalyst to speed up a reaction without being consumed by the reaction
The Activation Energy Barrier:
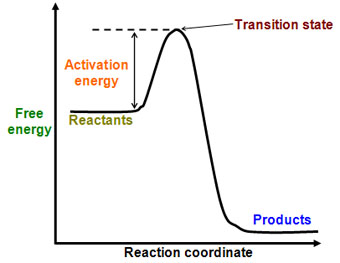
Activation energy: The initial energy investment required to start a reaction (energy required to contort the molecule into a highly unstable transition state so that the bonds may break)
-Think of the scenario of the ball on top of the hill again, this is the energy required to push the ball to the top of the hill, where is is unstable and likely to roll down the hill
- Activation energy is often supplied by heat. Heat increases the kinetic energy of reactant molecules, increasing the chance that they will collide and react
Look at the graph below, the peak in the graph represents the transition state and the energy needed to contort the molecule into its transition state (very high G). As atoms settle into more stable bonding arrangements, energy is released, resulting in products with a lower free energy (G).
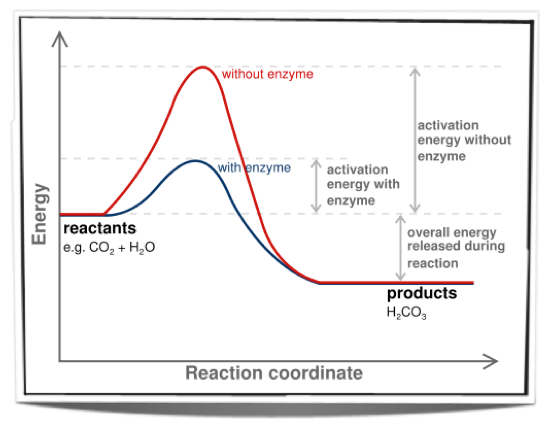
How Enzymes Speed Up Reactions:
-Living systems manage to maintain order, resisting the breakdown of macromolecules that have the potential to breakdown spontaneously because they cannot make it over the activation energy hump alone.
-Instead of using heat, since it could kill cells, denature proteins, an would speed up every reaction instead of select processes,
cells use catalysts (enzymes) to speed up reactions.
-Enzymes function as catalysts by lowering the activation energy needed for the reaction to proceed, allowing molecules to reach transitional states at even moderate temperatures
-enzymes cannot make a reaction that is endergonic exergonic, but can only speed up processes that would occur naturally.
Look at the graph below depicting how an enzyme can lower the activation energy necessary for an energetically favorable reaction to proceed.
-Living systems manage to maintain order, resisting the breakdown of macromolecules that have the potential to breakdown spontaneously because they cannot make it over the activation energy hump alone.
-Instead of using heat, since it could kill cells, denature proteins, an would speed up every reaction instead of select processes,
cells use catalysts (enzymes) to speed up reactions.
-Enzymes function as catalysts by lowering the activation energy needed for the reaction to proceed, allowing molecules to reach transitional states at even moderate temperatures
-enzymes cannot make a reaction that is endergonic exergonic, but can only speed up processes that would occur naturally.
Look at the graph below depicting how an enzyme can lower the activation energy necessary for an energetically favorable reaction to proceed.
Substrate Specificity of Enzymes:
An enzyme only works on specific substrates
-This specificity is due to the complex structure of an enzyme (protein) - think about primary through quarternary structure!
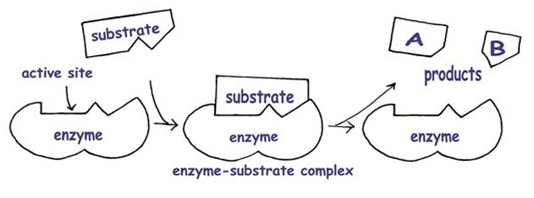
Substrate: the reactant an enzyme acts on
Enzyme-substrate complex: when an enzyme binds to its substrate/s
-While an enzyme is bound to its reactant/s the enzyme converts the reactant/s to the product/s
Active site: the region of the enzyme molecule that binds the substrate
-Typically a pocket or groove in structure that consists of only a few amino acids
-The rest of the enzyme, however, dictates the overall structure.
- The substrate is complementary to the active site of the enzyme, providing the specificity between the two.
Induced fit: An enzyme is not rigid in structure, but as the substrate enters the active site, the amino acid R groups interact with the substrate in a way that tightens the fit.
An enzyme only works on specific substrates
-This specificity is due to the complex structure of an enzyme (protein) - think about primary through quarternary structure!
Substrate: the reactant an enzyme acts on
Enzyme-substrate complex: when an enzyme binds to its substrate/s
-While an enzyme is bound to its reactant/s the enzyme converts the reactant/s to the product/s
Active site: the region of the enzyme molecule that binds the substrate
-Typically a pocket or groove in structure that consists of only a few amino acids
-The rest of the enzyme, however, dictates the overall structure.
- The substrate is complementary to the active site of the enzyme, providing the specificity between the two.
Induced fit: An enzyme is not rigid in structure, but as the substrate enters the active site, the amino acid R groups interact with the substrate in a way that tightens the fit.
Catalysis in the Enzyme's Active Site:
-in enzymatic reactions, the substrate is held in the active site by weak interactions (hydrogen or ionic bonds)
-the R groups of the enzyme's active site catalyze the reaction
-The product departs from the active site and the enzyme is left unchanged, free to take in another substrate molecule/s
-Most enzymes can catalyze the forward or reverse reaction depending on the direction that results in a negative ∆G, toward equilibrium
Mechanisms used by Enzymes to Lower Activation Energy (Speeding up a Reaction):
1) When there is more than one substrate, the active site arranges substrates in a way which orients them properly for the reaction to occur
2) The enzyme may stretch the substrate molecules toward transition state (highering their free energy (G)) making them more likely to breakdown.
3) The active site provides a microenvironment (within its pocket or groove) that is conducive to the reaction taking place
4) Amino acids within the active site actively participate in the reaction.
-Increasing substrate concentrations will increase the enzyme activity until the substrate reaches a concentration where the enzymes are all occupied (the enzymes are saturated) and no active sites are readily available.
-at this point the reaction rate cannot increase without an increase in the enzyme concentration
Effects of Local Conditions on Enzyme Activity:
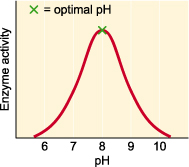
-Environmental conditions, such as pH and temperature, affect the activity of enzymes
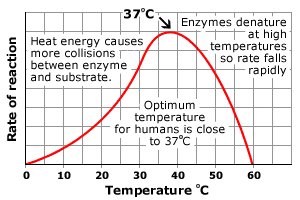
Effects of Temperature and pH:
-The temperature and pH at which an enzyme is most effective is called the optimal temperature (35-40*C) or optimal pH (6-8)
-With increasing temperature, up to a point, enzymatic action is increased
-However, as we know, proteins can be denatured at high temperatures
-If temperatures increase to where the proteins structure is compromised, the active site will be compromised, resulting in loss
of the enzyme's function.
-in enzymatic reactions, the substrate is held in the active site by weak interactions (hydrogen or ionic bonds)
-the R groups of the enzyme's active site catalyze the reaction
-The product departs from the active site and the enzyme is left unchanged, free to take in another substrate molecule/s
-Most enzymes can catalyze the forward or reverse reaction depending on the direction that results in a negative ∆G, toward equilibrium
Mechanisms used by Enzymes to Lower Activation Energy (Speeding up a Reaction):
1) When there is more than one substrate, the active site arranges substrates in a way which orients them properly for the reaction to occur
2) The enzyme may stretch the substrate molecules toward transition state (highering their free energy (G)) making them more likely to breakdown.
3) The active site provides a microenvironment (within its pocket or groove) that is conducive to the reaction taking place
4) Amino acids within the active site actively participate in the reaction.
-Increasing substrate concentrations will increase the enzyme activity until the substrate reaches a concentration where the enzymes are all occupied (the enzymes are saturated) and no active sites are readily available.
-at this point the reaction rate cannot increase without an increase in the enzyme concentration
Effects of Local Conditions on Enzyme Activity:
-Environmental conditions, such as pH and temperature, affect the activity of enzymes
Effects of Temperature and pH:
-The temperature and pH at which an enzyme is most effective is called the optimal temperature (35-40*C) or optimal pH (6-8)
-With increasing temperature, up to a point, enzymatic action is increased
-However, as we know, proteins can be denatured at high temperatures
-If temperatures increase to where the proteins structure is compromised, the active site will be compromised, resulting in loss
of the enzyme's function.
-Most enzymes have optimal pH's of between 6 and 8 and will be inhibited if the pH is changed from this range
-some enzymes that function in environments like the stomach, where the pH is acidic at 2, have optimal pH's that
are different and specific to their location
-some enzymes that function in environments like the stomach, where the pH is acidic at 2, have optimal pH's that
are different and specific to their location
Cofactors:
-Cofactors: nonprotein helpers that enhance an enzyme catalytic activity.
-cofactors may be pemanently bound or may bind loosely and reversibly along with the substrate.
-inorganic: Zinc, Iron, or Copper ions
-Coenzyme: an organic cofactor.
- vitamins are important for nutrition because they act as coenzymes or as materials that coenzymes are made of.
Enzyme Inhibitors:
-Competitive Inhibitors: reduce enzymatic activity by competing with substrates for active sites.
- can be overcome by increasing substrate concentration, and therefore the likelihood that the substrate will occupy the active site instead
-Noncompetitive Inhibitors: bind to another part (not the active site) of an enzyme, compromising the overall structure and therefore the active site.
Inhibitors may bind reversibly or irreversibly to an enzyme.
-toxins such as nerve gas, bind irreversibly.
-antibiotics bind to bacterial enzymes that catalyze the reactions necessary for cell wall production
The Evolution of Enzymes:
-Proteins are encoded by genes
-Mutations to genes can produce proteins that are different
-If the mutation enhances substrate/active site affinity, natural selection will promote the proliferation of the mutation
-If the mutation causes the affinity of a substrate different than the original substrate for the active site and it is a beneficial change, natural selection will promote the proliferation of the mutation
-If the mutation is not beneficial, than organisms with the mutation will die off.
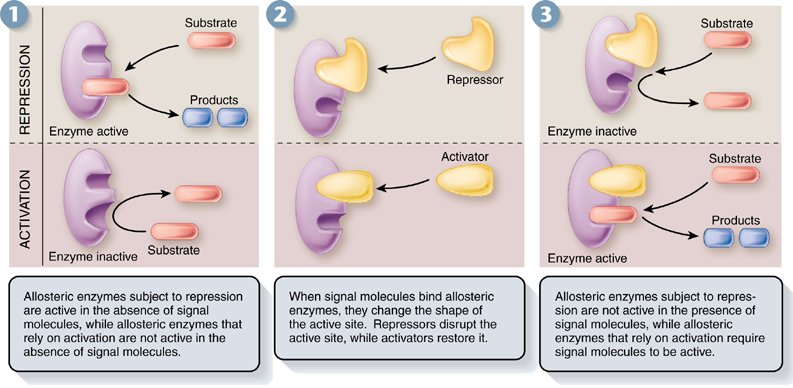
Allosteric Regulation of Enzymes:
Allosteric regulation: a protein's function at one site is affected by the binding of a regulatory molecule to a different site.
-can stimulate or inhibit enzyme activity
Allosteric Activation and Inhibition:
-Enzymes that are allosterically regulated usually consist of two or more subunits.
-If the regulator is inhibitory the binding of the regulator results in loss of active site structure, decreasing the affinity for the substrate
-If the regulator is stimulating the binding of the regulator results in a gain of active site structure, increasing the affinity for the substrate (cooperativity)
-Cofactors: nonprotein helpers that enhance an enzyme catalytic activity.
-cofactors may be pemanently bound or may bind loosely and reversibly along with the substrate.
-inorganic: Zinc, Iron, or Copper ions
-Coenzyme: an organic cofactor.
- vitamins are important for nutrition because they act as coenzymes or as materials that coenzymes are made of.
Enzyme Inhibitors:
-Competitive Inhibitors: reduce enzymatic activity by competing with substrates for active sites.
- can be overcome by increasing substrate concentration, and therefore the likelihood that the substrate will occupy the active site instead
-Noncompetitive Inhibitors: bind to another part (not the active site) of an enzyme, compromising the overall structure and therefore the active site.
Inhibitors may bind reversibly or irreversibly to an enzyme.
-toxins such as nerve gas, bind irreversibly.
-antibiotics bind to bacterial enzymes that catalyze the reactions necessary for cell wall production
The Evolution of Enzymes:
-Proteins are encoded by genes
-Mutations to genes can produce proteins that are different
-If the mutation enhances substrate/active site affinity, natural selection will promote the proliferation of the mutation
-If the mutation causes the affinity of a substrate different than the original substrate for the active site and it is a beneficial change, natural selection will promote the proliferation of the mutation
-If the mutation is not beneficial, than organisms with the mutation will die off.
- 8.5: Regulation of enzyme activity helps control metabolism
Allosteric Regulation of Enzymes:
Allosteric regulation: a protein's function at one site is affected by the binding of a regulatory molecule to a different site.
-can stimulate or inhibit enzyme activity
Allosteric Activation and Inhibition:
-Enzymes that are allosterically regulated usually consist of two or more subunits.
-If the regulator is inhibitory the binding of the regulator results in loss of active site structure, decreasing the affinity for the substrate
-If the regulator is stimulating the binding of the regulator results in a gain of active site structure, increasing the affinity for the substrate (cooperativity)
Feedback Inhibition:
Feedback Inhibition: a metabolic pathway is halted by the inhibitory binding of its end product to an enzyme that acts earlier on in the pathway.
-prevents wasted energy and accumulation of unnecessary product
Localization of Enzymes within the Cell:
-compartmentalization of the cell brings order to metabolic pathways
-Some enzymes are organized into multienzyme complexes to facilitate the sequence of reactions (the product of the first reaction serving as the substrate in the next)
Feedback Inhibition: a metabolic pathway is halted by the inhibitory binding of its end product to an enzyme that acts earlier on in the pathway.
-prevents wasted energy and accumulation of unnecessary product
Localization of Enzymes within the Cell:
-compartmentalization of the cell brings order to metabolic pathways
-Some enzymes are organized into multienzyme complexes to facilitate the sequence of reactions (the product of the first reaction serving as the substrate in the next)