Chapter 7: Membrane Structure and Function
Life at the Edge
-The plasma membrane serves at the boundary between a cell and its external environment, and controls the movement of substances into and out of the cell
Selective permeability: the constitution of the plasma membrane permits some substances to cross more easily than others
-Phospholipids are the most abundant lipids, and form a bilayer
-a bilayer has the hydrophilic heads on both sides with the hydrophobic tails in between.
Amphipathic: a molecule that contains opposing properties
-In the case of phospholipids, this refers to the molecules hydrophilic head regions and the hydrophobic tail regions.
-Membrane proteins are also amphipathic, their hydrophilic amino acid residues are found on the interior and exterior
of the membrane, while their hydrophobic residues are found in the hydrophobic region of the bilayer
Fluid Mosaic model: proteins form a mosaic within a fluid bilayer of phospholipids
-Proteins have distinct locations for efficiency of functions
The Fluidity of Membranes
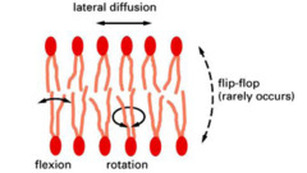
-hydrophobic interactions are responsible for keeping the cell membrane together.
- These are weak forces that permit lateral movement of both proteins and phospholipids.
- lipids move fast and frequently within their side of the bilayer
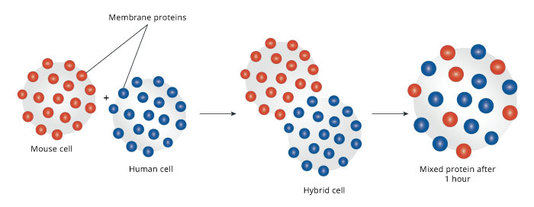
- proteins can move, but move more slowly (proven by Frye and Edinin)
-some proteins move specifically by interacting with the cytoskeleton's motor proteins
- Though infrequent, phospholipids can jump leaflets (sides of the bilayer)
-Some proteins bound to cytoskeleton or ECM attachments are immobile
Life at the Edge
-The plasma membrane serves at the boundary between a cell and its external environment, and controls the movement of substances into and out of the cell
Selective permeability: the constitution of the plasma membrane permits some substances to cross more easily than others
- 7.1: Cellular membranes are fluid mosaics of lipids and proteins
-Phospholipids are the most abundant lipids, and form a bilayer
-a bilayer has the hydrophilic heads on both sides with the hydrophobic tails in between.
Amphipathic: a molecule that contains opposing properties
-In the case of phospholipids, this refers to the molecules hydrophilic head regions and the hydrophobic tail regions.
-Membrane proteins are also amphipathic, their hydrophilic amino acid residues are found on the interior and exterior
of the membrane, while their hydrophobic residues are found in the hydrophobic region of the bilayer
Fluid Mosaic model: proteins form a mosaic within a fluid bilayer of phospholipids
-Proteins have distinct locations for efficiency of functions
The Fluidity of Membranes
-hydrophobic interactions are responsible for keeping the cell membrane together.
- These are weak forces that permit lateral movement of both proteins and phospholipids.
- lipids move fast and frequently within their side of the bilayer
- proteins can move, but move more slowly (proven by Frye and Edinin)
-some proteins move specifically by interacting with the cytoskeleton's motor proteins
- Though infrequent, phospholipids can jump leaflets (sides of the bilayer)
-Some proteins bound to cytoskeleton or ECM attachments are immobile
-Membranes must remain fluid to function
-permeability is altered if fluidity is altered
-proteins cannot move where they are needed if fluidity is altered
-Organisms that live in extreme environments have evolutionary adaptations that maintain the
appropriate amount of membrane fluidity even at extreme temperatures.
-Factors that affect fluidity:
1) temperature:
-increased temperatures increase fluidity
-decreased temperatures decrease fluidity
2) saturation of hydrocarbon tails:
-saturated phospholipids are less fluid (straight tails lead to tighter packing of phospholipids)
-unsaturated phospholipids are more fluid (kinks in tails, allow more movement)
3) presence of cholesterol: "fluidity buffer" system
-at high temperatures, cholesterol restricts phospholipid movement, making them less fluid
-at moderate temperatures, cholesterol permits more movement because it spaces out the packing of phospholipids, making them more fluid
Membrane Proteins and Their Functions:
-Membrane protein composition varies by cell type and then by organelle type within the cells
-These proteins can be secured in the extracellular environment by the extracellular matrix fibers or on the cytoplasmic side of the membrane by the cytoskeleton.
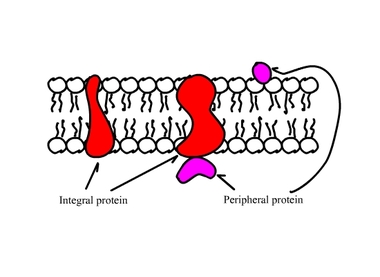
-Integral Protein: proteins embedded in cell membrane.
- Transmembrane integral proteins spans the entire width of the membrane.
- Other integral proteins only extended partially into the membrane.
-Portions of integral proteins embedded in bilayer consist of non-polar amino acids,
usually arranged in alpha helices.
-Some transmembrane proteins have hydrophilic "channels" that provide a pathway
for polar molecules through the membrane.
-Peripheral Protein: not embedded in bilayer, bound to surface of membrane or to an integral protein.
-permeability is altered if fluidity is altered
-proteins cannot move where they are needed if fluidity is altered
-Organisms that live in extreme environments have evolutionary adaptations that maintain the
appropriate amount of membrane fluidity even at extreme temperatures.
-Factors that affect fluidity:
1) temperature:
-increased temperatures increase fluidity
-decreased temperatures decrease fluidity
2) saturation of hydrocarbon tails:
-saturated phospholipids are less fluid (straight tails lead to tighter packing of phospholipids)
-unsaturated phospholipids are more fluid (kinks in tails, allow more movement)
3) presence of cholesterol: "fluidity buffer" system
-at high temperatures, cholesterol restricts phospholipid movement, making them less fluid
-at moderate temperatures, cholesterol permits more movement because it spaces out the packing of phospholipids, making them more fluid
Membrane Proteins and Their Functions:
-Membrane protein composition varies by cell type and then by organelle type within the cells
-These proteins can be secured in the extracellular environment by the extracellular matrix fibers or on the cytoplasmic side of the membrane by the cytoskeleton.
-Integral Protein: proteins embedded in cell membrane.
- Transmembrane integral proteins spans the entire width of the membrane.
- Other integral proteins only extended partially into the membrane.
-Portions of integral proteins embedded in bilayer consist of non-polar amino acids,
usually arranged in alpha helices.
-Some transmembrane proteins have hydrophilic "channels" that provide a pathway
for polar molecules through the membrane.
-Peripheral Protein: not embedded in bilayer, bound to surface of membrane or to an integral protein.
Six Important Functions of Membrane Proteins:
1) Transport: transmembrane proteins can provide channels or change shape to allow for the entry of specific solutes.
2) Enzyme Activity: some embedded proteins act as enzymes with their active site/s on the cytoplasmic side, and can be used to carry out several steps of metabolic pathways.
3) Signal Transduction: surface proteins can act as receptors for chemical messengers and relay the message to another protein within the cell.
4) Cell-Cell Recognition: glycoproteins (proteins with covalently bound carbohydrate chains) serve as "ID tags" for cell to cell recognition.
5) Intercellular Joining: proteins main link together to connect cells (junctions)
6) Attachment to the Cytoskeleton and Extracellular Matrix: proteins may bind noncovalently to fibers in cytoskeleton or extracellular matrix to increase cell's structural stability, communicate signals from internal environment to external environment or visa versa, or to anchor proteins in place.
The Role of Membrane Carbohydrates in Cell-Cell Recognition:
Cell-Cell Recognition: cell's ability to distinguish one type of neighboring cell from another
-Embryonic cell differentiation.
-Rejection of foreign cells by the immune system.
-Glycolipids and Glycoproteins contain covalently bound carbohydrate chains on the EC side of the membrane that are usually short and branched.
Synthesis and Sidedness of Membranes:
-Membranes inner and outer surfaces are distinct in composition.
-Directionality of constituents is determined as membrane by the ER and Golgi.
-The cell membrane's hydrophobic interior accounts for its selective permeability.
-Small, non-polar/hydrophobic molecules, like CO2 and O2, can easily diffuse through the membrane
-Large molecules, polar/hydrophilic molecules, or charged ions are slow to pass or required assistance
-Charged ions are the least agreeable
Transport Proteins:
-Membrane proteins also account for selective permeability.
-Proteins are very specific for which molecule or group of molecules they transport.
-Transport proteins span the membrane and can provide polar molecules and charged ions a pathway through the membrane
Channel Proteins: provide hydrophilic channel for passage of molecules
-Aquaporin: channel proteins responsible for letting water pass through the membrane (kidney cells/red blood cells)
Carrier Proteins: bind molecules and change shape, guiding them through hydrophobic portion of membrane.
-Glucose transport
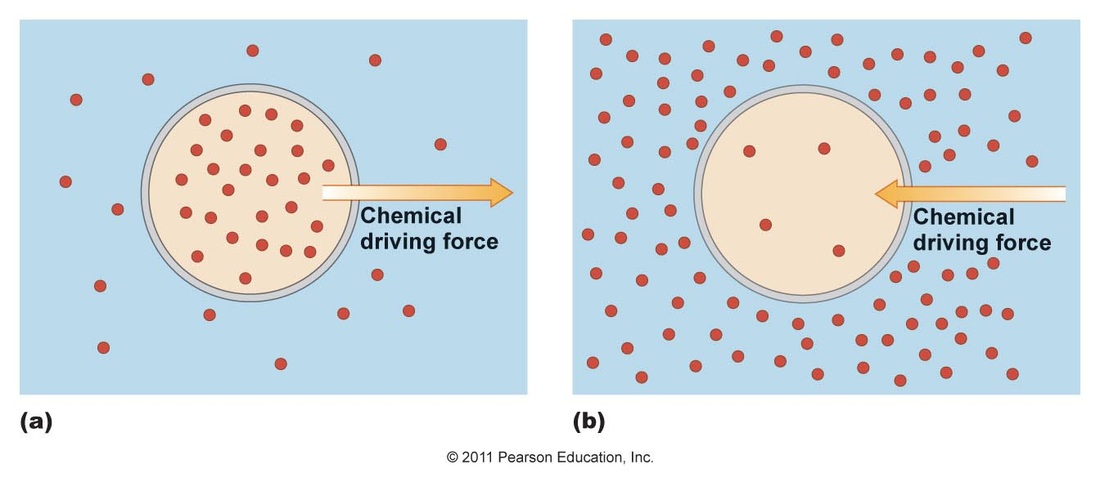
Diffusion: Type of passive transport where a substance moves across a semi-permeable membrane from an area of higher solute concentration to an area of lower solute concentration (down its concentration gradient).
- When the concentration of solute is equal on each side, the concentrations will remain in dynamic equilibrium with no net gain or loss to either side.
Effects of Osmosis on Water Balance:
Osmosis: the diffusion of free water across a semi-permeable membrane from an area of higher solute concentration to an area of lower solute concentration to even the concentrations of the solutions on both sides of the membrane.
-free water refers to water that is available to cross the membrane.
-some water may be bound to hydrophilic solutes, and therefore, unavailable.
Water Balance of Cells With and Without Cell Walls:
Tonicity: the ability of a surrounding solution to make a cell lose or gain water. Cell experience no net gain/loss of water (water move back and forth between EC space and cell at equal rates)
-dependent on concentrations of non-penetrating solutes in the EC environment
Isotonic Solutions: the concentration of solute is the same in EC space as it is in the cell.
-Animal: No changes to cell
-Plant: Since no net gain, plant cells become flaccid (limp)
Hypertonic Solutions: the concentration of solute is greater in the EC space than inside the cell. Cells experience net loss of water to EC environment.
-Animal cells shrivel
-Plant cell will shrink, membrane pulls away from cell wall, called plasmolysis, plant will wilt
-Bacterial cells with cell walls will also plasmolyze in hypertonic environments
Hypotonic Solutions:the concentration of solute is greater inside the cell than in the EC space. Cells experience net gain of water from EC environment.
-Animal cells swell and burst (lyse)
-Plant cells, beneficial because cells become turgid (firm), cell wall exerts turgor pressure that prevents excessive uptake of water.
Osmoregulation: control of solute concentrations and water balance
-Organisms that live in hypertonic or hypotonic environments have adaptations that allow them to osmoregulate.
-Unicellular, water-dwelling organisms may have contractile vacuoles to pump out water taken up by osmosis and prevent lysing of cell or cell membranes that are less permeable to water to slow the uptake.
Facilitated Diffusion: Passive Transport Aided by Proteins:
Facilitated Diffusion: polar molecules and ions diffuse passively across the membrane with the help of transport proteins, by proceeding down the gradient
(see transport proteins above)
Ion channels: channel proteins that transport ions
-Gated ion channels: many ion channels are gated and open or close in response to a stimulus.
-Stimulus may be electrical or another molecule binding to the transport protein
-Nervous system: electrical stimulus opens channel allowing K+ ions to flood in, this allows the nerve cell
to "reload" in order to fire again.
Active Transport: the energy-requiring, protein-facilitated movement of solutes across the membrane against their concentration gradients
- allows cell to maintain concentrations of small solutes that differ from the concentration in the EC space.
-ATP driven process
How Ion Pumps Work:
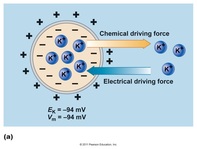
Two forces determine the movement of ions across the membrane:
1) chemical: the concentration gradient of each ion individually
2) electrical: membrane potential- the net concentration of all ions in the EC space and within the cell.
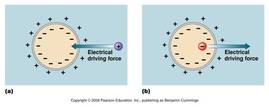
-Membrane potential: the voltage that exists across the plasma membrane of all cells, caused by uneven distribution of anions and cations on either side of the membrane, resulting in a net negative charge inside the cell and net positive charge in EC space.
- this potential affects the movement of ions across the membrane
-based on distribution, favors letting cations into cell and anions out of cell
-Together these forces make up the electrochemical gradient and determine whether ion movement is with or against the gradient, and therefore, whether movement requires energy or does not require energy
-If the two gradients favor the same direction of movement, no energy is required.
-If the two gradients favor opposite directions of movement, energy is required.
1) Transport: transmembrane proteins can provide channels or change shape to allow for the entry of specific solutes.
2) Enzyme Activity: some embedded proteins act as enzymes with their active site/s on the cytoplasmic side, and can be used to carry out several steps of metabolic pathways.
3) Signal Transduction: surface proteins can act as receptors for chemical messengers and relay the message to another protein within the cell.
4) Cell-Cell Recognition: glycoproteins (proteins with covalently bound carbohydrate chains) serve as "ID tags" for cell to cell recognition.
5) Intercellular Joining: proteins main link together to connect cells (junctions)
6) Attachment to the Cytoskeleton and Extracellular Matrix: proteins may bind noncovalently to fibers in cytoskeleton or extracellular matrix to increase cell's structural stability, communicate signals from internal environment to external environment or visa versa, or to anchor proteins in place.
The Role of Membrane Carbohydrates in Cell-Cell Recognition:
Cell-Cell Recognition: cell's ability to distinguish one type of neighboring cell from another
-Embryonic cell differentiation.
-Rejection of foreign cells by the immune system.
-Glycolipids and Glycoproteins contain covalently bound carbohydrate chains on the EC side of the membrane that are usually short and branched.
Synthesis and Sidedness of Membranes:
-Membranes inner and outer surfaces are distinct in composition.
-Directionality of constituents is determined as membrane by the ER and Golgi.
- 7.2: Membrane structure results in selective permeability:
-The cell membrane's hydrophobic interior accounts for its selective permeability.
-Small, non-polar/hydrophobic molecules, like CO2 and O2, can easily diffuse through the membrane
-Large molecules, polar/hydrophilic molecules, or charged ions are slow to pass or required assistance
-Charged ions are the least agreeable
Transport Proteins:
-Membrane proteins also account for selective permeability.
-Proteins are very specific for which molecule or group of molecules they transport.
-Transport proteins span the membrane and can provide polar molecules and charged ions a pathway through the membrane
Channel Proteins: provide hydrophilic channel for passage of molecules
-Aquaporin: channel proteins responsible for letting water pass through the membrane (kidney cells/red blood cells)
Carrier Proteins: bind molecules and change shape, guiding them through hydrophobic portion of membrane.
-Glucose transport
- 7.3: Passive transport is diffusion of a substance across a membrane with no energy investment
Diffusion: Type of passive transport where a substance moves across a semi-permeable membrane from an area of higher solute concentration to an area of lower solute concentration (down its concentration gradient).
- When the concentration of solute is equal on each side, the concentrations will remain in dynamic equilibrium with no net gain or loss to either side.
Effects of Osmosis on Water Balance:
Osmosis: the diffusion of free water across a semi-permeable membrane from an area of higher solute concentration to an area of lower solute concentration to even the concentrations of the solutions on both sides of the membrane.
-free water refers to water that is available to cross the membrane.
-some water may be bound to hydrophilic solutes, and therefore, unavailable.
Water Balance of Cells With and Without Cell Walls:
Tonicity: the ability of a surrounding solution to make a cell lose or gain water. Cell experience no net gain/loss of water (water move back and forth between EC space and cell at equal rates)
-dependent on concentrations of non-penetrating solutes in the EC environment
Isotonic Solutions: the concentration of solute is the same in EC space as it is in the cell.
-Animal: No changes to cell
-Plant: Since no net gain, plant cells become flaccid (limp)
Hypertonic Solutions: the concentration of solute is greater in the EC space than inside the cell. Cells experience net loss of water to EC environment.
-Animal cells shrivel
-Plant cell will shrink, membrane pulls away from cell wall, called plasmolysis, plant will wilt
-Bacterial cells with cell walls will also plasmolyze in hypertonic environments
Hypotonic Solutions:the concentration of solute is greater inside the cell than in the EC space. Cells experience net gain of water from EC environment.
-Animal cells swell and burst (lyse)
-Plant cells, beneficial because cells become turgid (firm), cell wall exerts turgor pressure that prevents excessive uptake of water.
Osmoregulation: control of solute concentrations and water balance
-Organisms that live in hypertonic or hypotonic environments have adaptations that allow them to osmoregulate.
-Unicellular, water-dwelling organisms may have contractile vacuoles to pump out water taken up by osmosis and prevent lysing of cell or cell membranes that are less permeable to water to slow the uptake.
Facilitated Diffusion: Passive Transport Aided by Proteins:
Facilitated Diffusion: polar molecules and ions diffuse passively across the membrane with the help of transport proteins, by proceeding down the gradient
(see transport proteins above)
Ion channels: channel proteins that transport ions
-Gated ion channels: many ion channels are gated and open or close in response to a stimulus.
-Stimulus may be electrical or another molecule binding to the transport protein
-Nervous system: electrical stimulus opens channel allowing K+ ions to flood in, this allows the nerve cell
to "reload" in order to fire again.
- 7.4: Active transport uses energy to move solutes against their concentration gradients
Active Transport: the energy-requiring, protein-facilitated movement of solutes across the membrane against their concentration gradients
- allows cell to maintain concentrations of small solutes that differ from the concentration in the EC space.
-ATP driven process
How Ion Pumps Work:
Two forces determine the movement of ions across the membrane:
1) chemical: the concentration gradient of each ion individually
2) electrical: membrane potential- the net concentration of all ions in the EC space and within the cell.
-Membrane potential: the voltage that exists across the plasma membrane of all cells, caused by uneven distribution of anions and cations on either side of the membrane, resulting in a net negative charge inside the cell and net positive charge in EC space.
- this potential affects the movement of ions across the membrane
-based on distribution, favors letting cations into cell and anions out of cell
-Together these forces make up the electrochemical gradient and determine whether ion movement is with or against the gradient, and therefore, whether movement requires energy or does not require energy
-If the two gradients favor the same direction of movement, no energy is required.
-If the two gradients favor opposite directions of movement, energy is required.
-Electrogenic Pumps: transport proteins that generates voltage across the membrane, helping to store energy that can be used for cellular work.
Animal Cells:
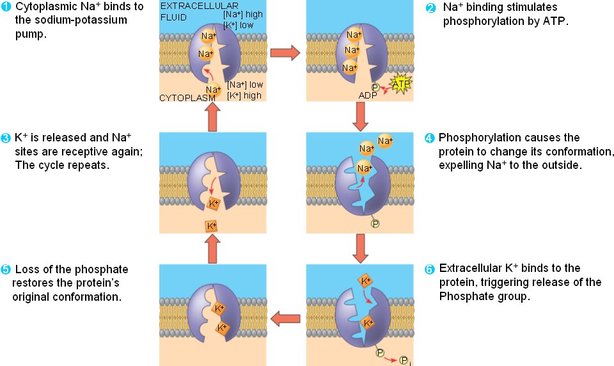
-Sodium-Potassium Pump: Pumps 3 Na+ ions out of cell in exchange for pumping 2 K+ ions into the cell.
-higher concentration of Na+ outside of the cell, and K+ inside cell
-pump works against both gradients
- contributes to membrane potential by pumping 3 cations out and only 2 cations in. (net 1+ to EC space)
Animal Cells:
-Sodium-Potassium Pump: Pumps 3 Na+ ions out of cell in exchange for pumping 2 K+ ions into the cell.
-higher concentration of Na+ outside of the cell, and K+ inside cell
-pump works against both gradients
- contributes to membrane potential by pumping 3 cations out and only 2 cations in. (net 1+ to EC space)
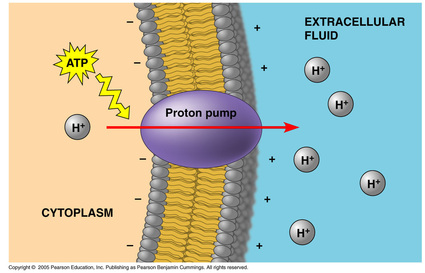
Plant, Fungi and Bacterial Cells:
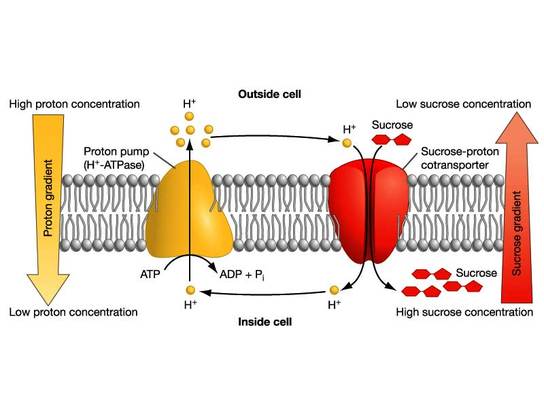
-Proton Pump: transports H+ ions out of cell.
-Proton Pump: transports H+ ions out of cell.
Cotransport: Coupled Transport by a Membrane Protein:
Cotransporter Proteins: couples diffusion of a substance along its gradient (maintained by electrogenic pumps) with the diffusion of another substance against its gradient.
-plants cotransport H+ with Sucrose
-animals cotransport Na+ with Glucose
Cotransporter Proteins: couples diffusion of a substance along its gradient (maintained by electrogenic pumps) with the diffusion of another substance against its gradient.
-plants cotransport H+ with Sucrose
-animals cotransport Na+ with Glucose
- 7.5: Bulk transport across the plasma membrane occurs by exocytosis and endocytosis
-The exit and entry of vesicles result in high turnover of membrane sections, keeping the membrane healthy
-The two processes are continuous, so there is no net loss or gain of membrane
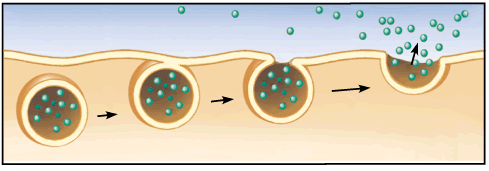
Exocytosis:
Exocytosis: secretion of molecules my fusion of a transport vesicle, carrying the substance for secretion, with the membrane.
-Vesicle produced from Golgi, moves along microtubules to exit cell.
-Vesicle fuses with plasma membrane and becomes part of membrane, spills contents into EC space
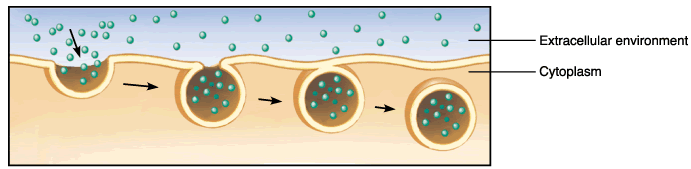
Endocytosis:
Endocytosis: the cell takes in molecules by forming a vesicle out of the membrane
-Membrane continues to sink inward until surrounds substance
-Pinches off as vesicle inside of cell
Three types:
1)Phagocytosis: cellular eating
2)Pinocytosis: cellular drinking
3)Receptor-mediated endocytosis: form of pinocytosis
-ligand: molecules that bind to specific receptors on membrane
-cells take in cholesterol using receptor-mediated endocytosis.
-low-density lipoproteins that carry cholesterol in the blood, bind LDL receptors on the cell membrane surface and enter the cell by endocytosis
-hypercholesterolemia occurs when LDL receptors are missing or do not function due to inherited mutation, results in high cholesterol levels in the blood and contribute to early development or atherosclerosis.
Endocytosis: the cell takes in molecules by forming a vesicle out of the membrane
-Membrane continues to sink inward until surrounds substance
-Pinches off as vesicle inside of cell
Three types:
1)Phagocytosis: cellular eating
2)Pinocytosis: cellular drinking
3)Receptor-mediated endocytosis: form of pinocytosis
-ligand: molecules that bind to specific receptors on membrane
-cells take in cholesterol using receptor-mediated endocytosis.
-low-density lipoproteins that carry cholesterol in the blood, bind LDL receptors on the cell membrane surface and enter the cell by endocytosis
-hypercholesterolemia occurs when LDL receptors are missing or do not function due to inherited mutation, results in high cholesterol levels in the blood and contribute to early development or atherosclerosis.