Chemistry Concepts:
1. Elements vs. Compounds
1. Elements vs. Compounds
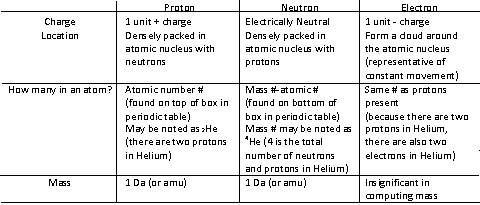
2. Subatomic Particles and Properties
Miscellaneous:
- The number of protons distinguishes an element's identity.
-All atoms are neutral in charge unless otherwise specified bc of even number of electrons and protons
-Only electrons in the valence shell of an atom participate in chemical bonds and reactions.
3. Atomic Number, Mass and Weight
Atomic #: The number of protons, which is unique to each element. May be written as a subscript as indicated in the chart above.
Atomic Mass: Mass of the nucleus, since protons and electrons account for the mass in an atom. The average of all the element's naturally occurring isotopes, weighted according to their abundance in nature. (g/mol)
Atomic Weight: Mass of one mole. A mole is simply a unit of measurement. Units are invented when existing units are inadequate. Chemical reactions often take place at levels where using grams wouldn't make sense, yet using absolute numbers of atoms/molecules/ions would be confusing, too. Like all units, a mole has to be based on something reproducible. A mole is the quantity of anything that has the same number of particles found in 12.000 grams of carbon-12. That number of particles is Avogadro's Number, which is roughly 6.02x1023. A mole of carbon atoms is 6.02x1023carbon atoms.
Mass number: The sum of the number of protons and neutrons present, assuming that each proton and neutron has a weight of 1 Da or amu each.
4. Isotopes
Isotope: different atomic forms of the same element, differing in the number of neutrons only.
-Elements occur naturally in a mixture of isotopes
-All isotopes have slightly different masses (more neutrons=larger mass, less neutrons=smaller mass)
-The atomic masses listed on the periodic table are averages of all the isotopes of that atom that exist in
nature, weighted according to their abundance.
-To calculate average atomic mass (if you aren't given which element you are working with, you may use
this to determine it based on your answer, using your periodic table).
(abundance of isotope A-as a decimal) (relative atomic mass of isotope A) + (abundance of isotope B-as a decimal) (relative atomic mass of isotope B)....
5. Electron Configuration Rules:
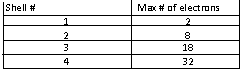
-Electrons will first occupy the shell with the lowest potential energy to capacity prior to filling the next shell.
-(Generally )
- The number of protons distinguishes an element's identity.
-All atoms are neutral in charge unless otherwise specified bc of even number of electrons and protons
-Only electrons in the valence shell of an atom participate in chemical bonds and reactions.
3. Atomic Number, Mass and Weight
Atomic #: The number of protons, which is unique to each element. May be written as a subscript as indicated in the chart above.
Atomic Mass: Mass of the nucleus, since protons and electrons account for the mass in an atom. The average of all the element's naturally occurring isotopes, weighted according to their abundance in nature. (g/mol)
Atomic Weight: Mass of one mole. A mole is simply a unit of measurement. Units are invented when existing units are inadequate. Chemical reactions often take place at levels where using grams wouldn't make sense, yet using absolute numbers of atoms/molecules/ions would be confusing, too. Like all units, a mole has to be based on something reproducible. A mole is the quantity of anything that has the same number of particles found in 12.000 grams of carbon-12. That number of particles is Avogadro's Number, which is roughly 6.02x1023. A mole of carbon atoms is 6.02x1023carbon atoms.
Mass number: The sum of the number of protons and neutrons present, assuming that each proton and neutron has a weight of 1 Da or amu each.
4. Isotopes
Isotope: different atomic forms of the same element, differing in the number of neutrons only.
-Elements occur naturally in a mixture of isotopes
-All isotopes have slightly different masses (more neutrons=larger mass, less neutrons=smaller mass)
-The atomic masses listed on the periodic table are averages of all the isotopes of that atom that exist in
nature, weighted according to their abundance.
-To calculate average atomic mass (if you aren't given which element you are working with, you may use
this to determine it based on your answer, using your periodic table).
(abundance of isotope A-as a decimal) (relative atomic mass of isotope A) + (abundance of isotope B-as a decimal) (relative atomic mass of isotope B)....
5. Electron Configuration Rules:
-Electrons will first occupy the shell with the lowest potential energy to capacity prior to filling the next shell.
-(Generally )
- An atom's electron configuration is a numeric representation of its electron orbitals. Electron orbitals are differently-shaped regions around an atom's nucleus where electrons are mathematically likely to be located. An electron configuration can quickly and simply tell you how many electron orbitals an atom has as well as the number of electrons populating each of its orbitals.
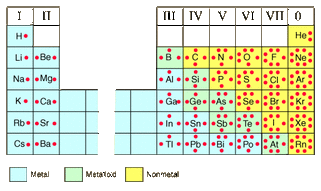
-To determine the number of electrons present in an atom, refer to the atomic # (# of protons, located on the top of the boxes in the periodic table), there are as many electrons as there are protons. You can do the math using this number to determine how many electrons will be present in the valence (outermost) shell by using the table above or, more simply, using the column #'s on the periodic table ( though this method does not always apply to transition metals).
- The Pauli exclusion principle states no two electrons can have the identical quantum mechanical state in the same atom, cannot be moving in the exact same space, this governs electron placement in orbitals (there can be multiple orbitals in each shell).
-The Octet Rule: General rule that states that atoms, in forming compounds, will gain, lose or share electrons to achieve a valence shell that mirrors that of the stable and noble gases, with a valence of 8 electrons (column 18 elements).
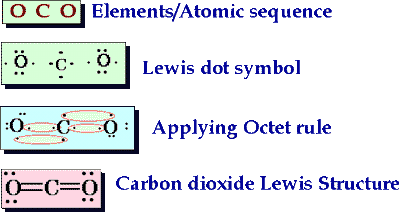
6. & 7. Lewis Dot Structures for Elements and Molecules
-Lewis dot structures only represent the valence shell of electrons in each atom
-We use black dots around the element's symbol to represent these electrons, the electrons should be distributed as evenly as possible, with a maximum of two on each side, UNLESS we are sharing electrons in double or triple covalent bonds.
-When working with molecules, draw each individual lewis dot structure to help yourself visually. Ask yourself, which shell is my valence shell, and how many electrons are there in it. Now, based on the octet rule (unless the valence shell is the only shell, like in Hydrogen) how many more electrons do you need to fill this shell to capacity.
Arrange the electrons between the the atoms present so that all of their shells are full.
Don't forget to ask yourself, HOW many Bonds can I form to help yourself place atoms within your structure? (Generally) Atoms can only form as many bonds as electrons they are missing from reaching an octet in their valence shell.
-To determine the number of electrons present in an atom, refer to the atomic # (# of protons, located on the top of the boxes in the periodic table), there are as many electrons as there are protons. You can do the math using this number to determine how many electrons will be present in the valence (outermost) shell by using the table above or, more simply, using the column #'s on the periodic table ( though this method does not always apply to transition metals).
- The Pauli exclusion principle states no two electrons can have the identical quantum mechanical state in the same atom, cannot be moving in the exact same space, this governs electron placement in orbitals (there can be multiple orbitals in each shell).
-The Octet Rule: General rule that states that atoms, in forming compounds, will gain, lose or share electrons to achieve a valence shell that mirrors that of the stable and noble gases, with a valence of 8 electrons (column 18 elements).
6. & 7. Lewis Dot Structures for Elements and Molecules
-Lewis dot structures only represent the valence shell of electrons in each atom
-We use black dots around the element's symbol to represent these electrons, the electrons should be distributed as evenly as possible, with a maximum of two on each side, UNLESS we are sharing electrons in double or triple covalent bonds.
-When working with molecules, draw each individual lewis dot structure to help yourself visually. Ask yourself, which shell is my valence shell, and how many electrons are there in it. Now, based on the octet rule (unless the valence shell is the only shell, like in Hydrogen) how many more electrons do you need to fill this shell to capacity.
Arrange the electrons between the the atoms present so that all of their shells are full.
Don't forget to ask yourself, HOW many Bonds can I form to help yourself place atoms within your structure? (Generally) Atoms can only form as many bonds as electrons they are missing from reaching an octet in their valence shell.
8. Electrons and bonding:
-Electrons in the valence shell of an atom are the only subatomic particles that will participate in bonding.
-Electronegativity is basically an atom's ability to attract electrons
-based on # of protons in nucleus, an atom with more protons (higher +) will have more pull on negatively
charged electrons
-Also based on the distance of the valence shell from the nucleus, further shells will not have as much "pull"
exerted on them by the nucleus
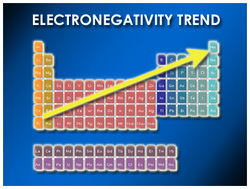
-electronegativity increasing from left to right and from bottom to top when you are looking at the periodic table.
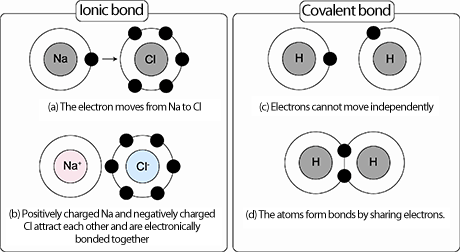
-The more electronegative atom in a covalent bond will have a greater share of the electrons being shared, in an ionic bond, the more electronegative atom "steals" the electron/s and becomes a negatively charged anion to the - x (x being the # of electrons gained) degree, while the less electronegative atom that "gives up" the electron/s becomes a positively charged cation to the +x (x being the number of electrons lost) degree
-The most electronegative elements are Fluorine, Oxygen, Chlorine, and Nitrogen, in
that order (following periodic trends, of course, if you forget!)
-Electrons in the valence shell of an atom are the only subatomic particles that will participate in bonding.
-Electronegativity is basically an atom's ability to attract electrons
-based on # of protons in nucleus, an atom with more protons (higher +) will have more pull on negatively
charged electrons
-Also based on the distance of the valence shell from the nucleus, further shells will not have as much "pull"
exerted on them by the nucleus
-electronegativity increasing from left to right and from bottom to top when you are looking at the periodic table.
-The more electronegative atom in a covalent bond will have a greater share of the electrons being shared, in an ionic bond, the more electronegative atom "steals" the electron/s and becomes a negatively charged anion to the - x (x being the # of electrons gained) degree, while the less electronegative atom that "gives up" the electron/s becomes a positively charged cation to the +x (x being the number of electrons lost) degree
-The most electronegative elements are Fluorine, Oxygen, Chlorine, and Nitrogen, in
that order (following periodic trends, of course, if you forget!)
Polar Bear and Penguin models for understanding polarity:
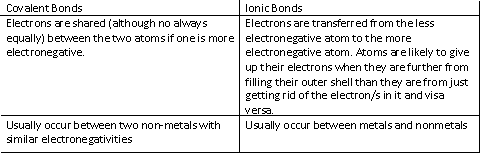
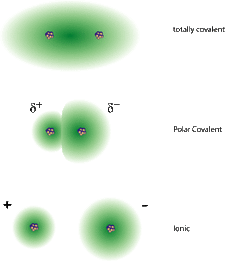
9. Covalent vs Ionic Bonds
-Covalent bonds can be single, double or triple bonds, depending on the number of electrons required for each atom to be satisfied. They may have to share 2 electrons (single bond), 4 electrons (double bond), or 6 electrons (triple bond)
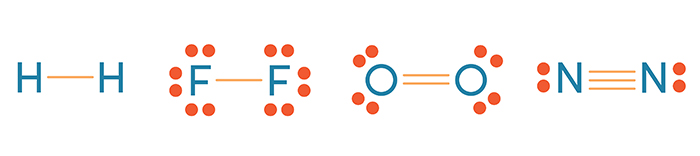
- a good example of this would be our diatomic elements, H2, Br2, F2, Cl2, O2, N2, I2
- These elements are not present as single atoms in nature, they only exist bound to themselves:
- a good example of this would be our diatomic elements, H2, Br2, F2, Cl2, O2, N2, I2
- These elements are not present as single atoms in nature, they only exist bound to themselves:
10. Polar vs Non-polar
- A molecule is polar if the shared electrons of their covalent bond are not shared equally, giving a partially negative charge to the atom holding the electrons closer, and a partially positive charge to the atom that is not. These partially charged regions are called dipoles.
- Non-polar molecules share their electrons evenly, and are not partially charged.
- A molecule is polar if the shared electrons of their covalent bond are not shared equally, giving a partially negative charge to the atom holding the electrons closer, and a partially positive charge to the atom that is not. These partially charged regions are called dipoles.
- Non-polar molecules share their electrons evenly, and are not partially charged.
As Far as solutions are concerned. There is a rule that governs whether or not two substances will mix or separate.
- Like dissolves like is the rule:
Polar substances WILL dissolve in Polar substances
Non-polar substances WILL dissolve in nonpolar substances
Non-polar substances WILL NOT dissolve in polar substances
Polar substances WILL NOT dissolve in nonpolar substances
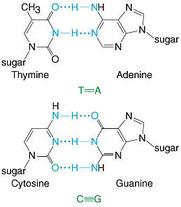
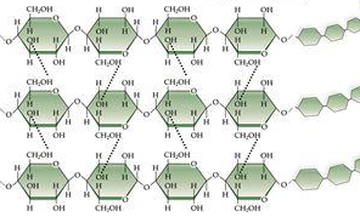
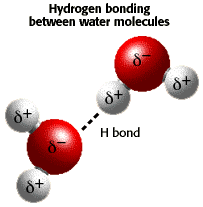
11. Hydrogen Bonds
- A hydrogen bond refers to the electromagnetic attraction between two polar molecules, where there is a Hydrogen attached to a much more electronegative atom. It is not a physical bond, it is just an attraction between dipoles. They are individually weak, but strong in high numbers. They are represented as dashed lines. ( below are pictures of hydrogen bonds that occur in DNA and Cellulose, and between water molecules)
- Like dissolves like is the rule:
Polar substances WILL dissolve in Polar substances
Non-polar substances WILL dissolve in nonpolar substances
Non-polar substances WILL NOT dissolve in polar substances
Polar substances WILL NOT dissolve in nonpolar substances
11. Hydrogen Bonds
- A hydrogen bond refers to the electromagnetic attraction between two polar molecules, where there is a Hydrogen attached to a much more electronegative atom. It is not a physical bond, it is just an attraction between dipoles. They are individually weak, but strong in high numbers. They are represented as dashed lines. ( below are pictures of hydrogen bonds that occur in DNA and Cellulose, and between water molecules)
12. Hydrophobic and Hydrophilic
-Hydrophobic: water repelling, molecules that repel water and do not mix with it (non polar or uncharged atoms)
-Hydrophilic: water loving, molecules that attract water and mix well with it (polar molecules or ions)
-molecules may have both hydrophilic and hydrophobic regions. (i.e. the phosopholipid bilayer that composes the cell membrane)
13. Acids and Bases
-Acids: compounds that release hydrogen ions (H+) in solution, referred to as proton donors bc H without it's electron is just a proton.
-Bases: compounds that release hydroxide (OH-) ions in solution, or can accept a hydrogen ion, proton acceptors.
-In solution together, acids and bases can neutralize each other, forming water molecules (H2O) and a salt.
-pH: Acids increase the concentration of H+ ions, while bases decrease the amount of H+ ions by accepting them. Acidity and basicity are therefore based on the concentration of H+ ions. (pH = -log [H+])
- the pH scale runs from 0 to 14, 0 being most acidic, and 14 being most basic.
14. Bonding with Carbon, Isomers
http://home.earthlink.net/~dayvdanls/CarbonChem.html
^Please look at me, I am just an abbreviated summary that will make your life easier.
-Hydrophobic: water repelling, molecules that repel water and do not mix with it (non polar or uncharged atoms)
-Hydrophilic: water loving, molecules that attract water and mix well with it (polar molecules or ions)
-molecules may have both hydrophilic and hydrophobic regions. (i.e. the phosopholipid bilayer that composes the cell membrane)
13. Acids and Bases
-Acids: compounds that release hydrogen ions (H+) in solution, referred to as proton donors bc H without it's electron is just a proton.
-Bases: compounds that release hydroxide (OH-) ions in solution, or can accept a hydrogen ion, proton acceptors.
-In solution together, acids and bases can neutralize each other, forming water molecules (H2O) and a salt.
-pH: Acids increase the concentration of H+ ions, while bases decrease the amount of H+ ions by accepting them. Acidity and basicity are therefore based on the concentration of H+ ions. (pH = -log [H+])
- the pH scale runs from 0 to 14, 0 being most acidic, and 14 being most basic.
14. Bonding with Carbon, Isomers
http://home.earthlink.net/~dayvdanls/CarbonChem.html
^Please look at me, I am just an abbreviated summary that will make your life easier.