- 9: Cellular Respiration and Fermentation
-Living systems require a constant input of energy to survive.
-organisms must take in more energy than they use
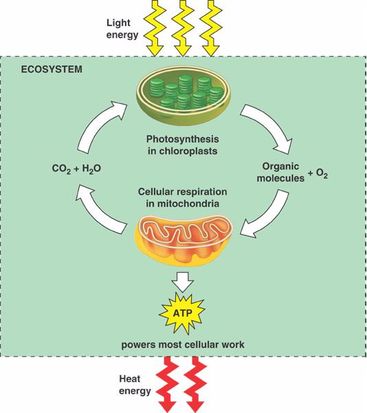
-Energy enters a system in the form of solar energy
-this solar energy drives the process of photosynthesis ( 6CO2 + 6H2O ---> C6H12O6 + 6O2 )
-The products of photosynthesis become the reactants for cellular respiration (C6H12O6 + 6O2 --->6CO2 + 6H2O)
-Energy leaves a system in the form of heat
-released from metabolic reactions and dissipates into surroundings
-Chemical Elements required for life are recycled
- 9.1: Catabolic Pathways and Production of ATP
Catabolic Pathways and Production of ATP:
-Organic compounds possess potential energy as a result of electron arrangement in their bonds
-Compounds that can be broken down in exergonic reactions, can supply energy for cellular work
Fermentation: the partial degradation of sugars or other organic fuel that occurs in the absence of O2 (anaerobic), less efficient in energy production
Aerobic Respiration: O2 consumed (aerobic respiration) as a reactant along with sugars, more efficient for energy production
Cellular respiration: the process in which organisms convert chemical energy into usable energy (ATP). Refers to both aerobic and anaerobic processes, but is usually used to refer to aerobic respiration.
Organic compounds + O2 ---> CO2 + H2O + Energy (ATP + heat)
The organic compounds, or "chemical energy" used in cellular respiration are most commonly carbohydrates and fats, but protein molecules can also be consumed as fuel.
-Starch is a major source of carbohydrates in animal diets. As you know, starch is composed
of glucose monomers, and it can be -hydrolyzed to free glucose molecules for cellular respiration.
-Glucose is then broken down via cellular respiration in a series of exergonic reactions.
-These exergonic reactions result in the phosphorylation of ADP, forming ATP, that can perform work.
-The exergonic reactions, themselves, do not perform work.
Redox Reactions: Oxidation and Reduction:
-The relocation of electrons releases energy stored in organic molecules, and this energy is used to synthesize ATP
The Principle of Redox
Redox Reactions: The transfer of electrons from one reactant to another.
-The reactant losing the electrons is said to become oxidized, and therefore the reducing agent (supplied the electrons)
-The reactant gaining the electrons is said to become reduced (more negative due to the gain of more electrons), and therefore the oxidizing agent (stole the electrons)
-Energy must be added to remove an electron from a reactant
-The more electronegative the atom, the harder it is to remove the electron, and the more energy it would require.
-An electron decreases in potential energy when it is transferred to a more electronegative atom from a less electronegative atom.
-The decrease in energy makes the redox reaction exergonic, and the energy can be used to do work.
-A complete transfer of electrons is not necessary for the reaction to be a redox reaction, it can include electrons being unequally shared in a covalent bond due to the differing electronegativities of the compounds atoms.
-Oxygen is one of the most electronegative atoms, and is therefore, a very potent oxidizing agent.
Oxidation of Organic Fuel Molecules During Cellular Respiration:
-Carbohydrates and fats have an abundance of hydrogen, making them the main energy yielding food sources
- the electrons in these molecules have high potential energy within their bonds, making them
perfect for traveling down the energy gradient of the electron transport chain.
-activation energy is the only thing preventing the electrons within these molecules from instantly
leaving to join oxygen.
-Therefore, the presence of enzymes control the reactions of cellular respiration.
Stepwise Energy Harvest via NAD+ and the Electron Transport Chain
-Food molecules are broken down slowly in a step-by-step fashion so that the energy released by each step may be used efficiently instead of wasted.
-Each of these steps are catalyzed by separate enzymes.
-Electrons removed in the form of hydrogen atoms from (for example) a molecule of glucose by dehydrogenases (glucose becomes oxidized), are first transferred to NAD+
-NAD+ is a coenzyme and electron carrier
-NAD+ reduces to NADH when it receives two electrons and 1 proton. The excess proton, H+, is released into solution.
-NADH is a good electron carrier bc electrons lose very little energy when they are transferred to NAD+
-NADH transports electrons to the electron transport chain, a series of proteins embedded in the inner membrane of the mitochondria
-as electrons move down the chain a series of redox reactions occur and the electrons proceed toward lower energy
as they are accepted by increasingly electronegative carriers in the chain.
-Oxygen is the final electron acceptor.
Summary of electron movement: Glucose ---> NAD+ ----> ETC ----> Oxygen
The Stages of Cellular Respiration
1) Glycolysis: Catabolic pathway that breaks down glucose into two molecules of pyruvate, occurs in cytoplasm
2) Pyruvate Oxidation and the Citric Acid Cycle (Krebs): pyruvate is oxidized in the mitochondrial matrix to acetyl coA, and acetyl coA enters citric acid cycle and carbon dioxide is released.
3) Oxidative Phosphorylation (ETC and Chemiosmosis): ETC accepts electrons from NADH and FADH2 (resulting from the first 2 steps), at the end of the chain oxygen accepts the electrons, and a molecule of water is formed. Hydrogen ions are pumped into the intermembrane space using the energy from the redox reactions of passed electrons, and this proton gradient powers ATP synthesis.
-Cell respiration technically refers to the release of CO2, so only steps 2 and 3 are included)
Oxidative Phosphorylation: synthesis of ATP from ADP and inorganic phosphate, powered by redox reactions of ETC (occurs in 3rd step only)
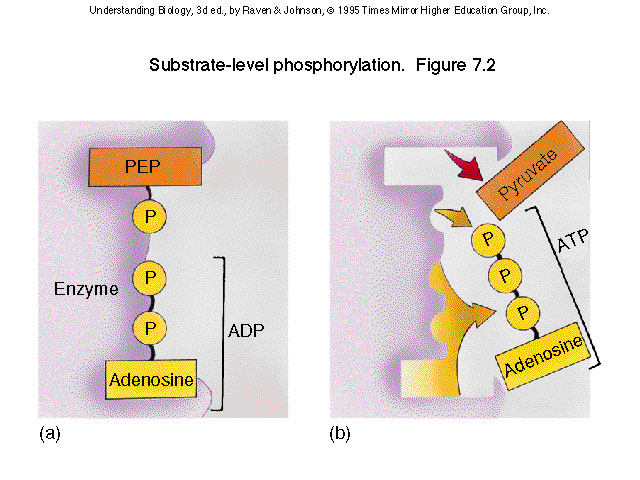
Substrate-level Phosphorylation: when ATP is synthesized by an enzyme transferring a phosphate group from a substrate molecule to ADP. (occurs in 1st and 2nd steps)
- 9.2: Glycolysis harvests chemical energy by oxidizing glucose to pyruvate:
1) 6-C glucose is split into two 3-C sugars
2) 3-C sugars oxidized, atoms rearranged to form 2 molecules of pyruvate
3) Pyruvate is ionized to become pyruvic acid
Results per Glucose molecule:
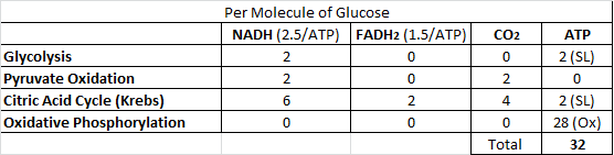
- NET GAIN 2 ATP (2 ATP used during process, 4 ATP formed)
-2 NADH (proceed to ETC)
- 9.3: After pyruvate is oxidized, the citric acid cycle completes the energy yielding oxidation of organic molecules
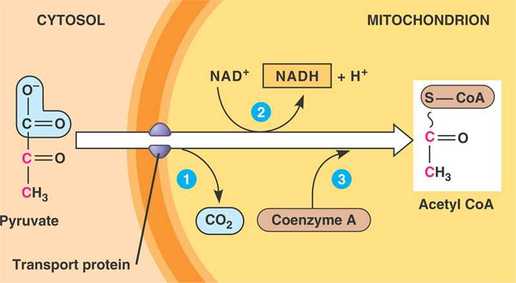
1) Pyruvate enters the mitochondrion via active transport
2) Pyruvate releases molecule of CO2
3) The remaining 2-C molecule is oxidized to acetate, the electrons are transferred to NAD+, forming NADH
4) The high energy acetyl CoA is then formed with the addition of coenzyme A to the 2-C molecules.
5) The acetyl group of these molecules enter the citric acid cycle
Results per glucose molecule:
- 2 CO2 molecules (byproduct)
-2 NADH molecules (proceed to ETC)
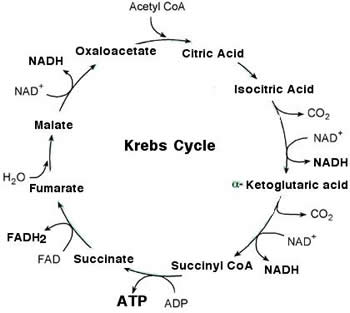
The Citric Acid Cycle aka Krebs Cycle
(1 complete cycle per acetyl coA molecule derived from the pyruvate molecules that were the product of glycolysis, so for a net product per Glucose molecule, the cycle will be repeated twice)
1) 2-C ace)tyl group enters cycle and combines with oxaloacetate to form citrate. Citrate is ionized to form citric acid (hence the citric acid cycle name)
2) Citrate is converted to an isomer, isocitrate by the removal of a water molecule
3) Isocitrate is oxidized, reducing NAD+ to NADH and the resulting compound loses a CO2 molecule, converting it to alpha-ketoglutarate
4) Alpha-ketoglutarate loses a molecule of CO2 and the resulting compound is oxidized by reducing another molecule of NAD+ to NADH and the product is combined with coA by an unstable bond, forming succinyl coA
5) Succinyl coA is removed by the addition of a phosphate group that is then transferred to a molecule of GDP to form GTP
(Guanosine-5'-triphosphate, a purine nucleoside triphosphate), GTP can then be used to phosphorylate ADP to ATP via substrate level phosphorylation. The resulting molecule is succinate.
6) Succinate is oxidized by reducing a molecule of FAD to FADH2, forming fumarate.
7) Fumarate gains a molecule of water and becomes Malate.
8) Malate is oxidized by reducing NAD+ to NADH, regenerating oxaloacetate.
Results per glucose molecule:
- 6 NADH (proceed to ETC)
- 2 FADH2 (proceed to ETC)
- 2 ATP
- 4 CO2 (byproduct)
(1 complete cycle per acetyl coA molecule derived from the pyruvate molecules that were the product of glycolysis, so for a net product per Glucose molecule, the cycle will be repeated twice)
1) 2-C ace)tyl group enters cycle and combines with oxaloacetate to form citrate. Citrate is ionized to form citric acid (hence the citric acid cycle name)
2) Citrate is converted to an isomer, isocitrate by the removal of a water molecule
3) Isocitrate is oxidized, reducing NAD+ to NADH and the resulting compound loses a CO2 molecule, converting it to alpha-ketoglutarate
4) Alpha-ketoglutarate loses a molecule of CO2 and the resulting compound is oxidized by reducing another molecule of NAD+ to NADH and the product is combined with coA by an unstable bond, forming succinyl coA
5) Succinyl coA is removed by the addition of a phosphate group that is then transferred to a molecule of GDP to form GTP
(Guanosine-5'-triphosphate, a purine nucleoside triphosphate), GTP can then be used to phosphorylate ADP to ATP via substrate level phosphorylation. The resulting molecule is succinate.
6) Succinate is oxidized by reducing a molecule of FAD to FADH2, forming fumarate.
7) Fumarate gains a molecule of water and becomes Malate.
8) Malate is oxidized by reducing NAD+ to NADH, regenerating oxaloacetate.
Results per glucose molecule:
- 6 NADH (proceed to ETC)
- 2 FADH2 (proceed to ETC)
- 2 ATP
- 4 CO2 (byproduct)
- 9.4: During oxidative phosphorylation, chemiosmosis couples electron transport to ATP synthesis
-The 10 NADH (2 from glycolysis, 2 from the oxidation of pyruvate and 6 from the citric acid cycle) and 2 FADH2 (both from the citric acid cycle) molecules produced by these cycles move toward the oxidative phosphorylation machinery.
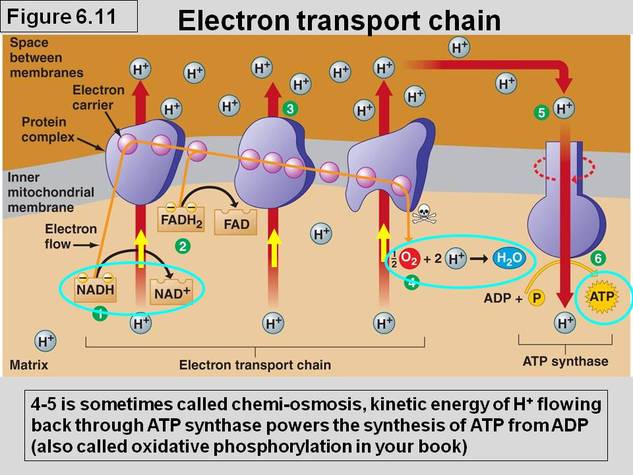
The Pathway of Electron Transport:
- The electron transport chain is a series of proteins embedded in the inner mitochondrial membrane.
- Due to the increased surface area provided by the cristae of this inner membrane, there are multiple ETCs in place.
-Most components of the ETC are protein complexes consisting of multiple proteins and are numbered I through IV.
-Electrons travel down the ETC, decreasing in potential energy as they are transferred to increasingly electronegative molecules along the chain.
-The molecule that loses the electrons returns to its oxidized state as it reduces the next electron transport molecule.
-Oxygen is the terminal electron acceptor of the ETC (the last molecule to be reduced), oxygen is notoriously very
electronegative.
NADH pathway:
1) NADH is oxidized to NAD+ by reducing the first molecule of complex I, flavoprotein.
2) Within complex I, the flavoprotein component passes the electrons to an iron-sulfur protein.
3) The iron-sulfur protein then passes its electrons to a ubiquinone (Q) molecule (the only non-protein member of the ETC) that does not exist within a complex, but as its own entity.
4) The Q molecule passes its electrons to the 1st cytochrome protein (proteins containing heme (Fe) groups) of the ETC in complex III, called cytochrome b.
5) Within complex III electrons are transferred from cytochrome b to another iron-sulfur protein and then to cytochrome c1.
6) From cytochrome c1, electrons are transferred to the mobile and separate entity cytochrome c.
7) Cytochrome c transports electrons to the cytochrome a of complex 4.
8) Within complex IV, electrons are transferred from cytochrome a to cytochrome a3.
9) Cytochrome a3 transfers electrons to and oxygen molecule (O2).
10) Each oxygen atom then picks up a pair of hydrogen atoms to form H2O.
FADH2 Pathway:
1) FADH2 is oxidized to FAD by reducing the first molecule in complex II, an iron-sulfur protein.
....Then follows steps 3-10 above
-This means that FADH2 enters the pathway at a lower point in the energy gradient than does NADH.
-So although both carriers donate 2 electrons to the chain each, FADH2 ultimately produces only 1/3 of the energy for ATP synthesis that NADH does.
THE ELECTRON TRANSPORT CHAIN ITSELF, DOES NOT PRODUCE ANY ATP!!
For your enjoyment and helpful for memorization: THE ETC RAP- What whhhhat!
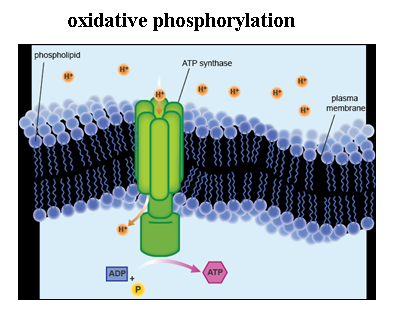
Chemiosmosis: The Energy-Coupling Mechanism
- Although the ETC does not produce ATP on its own, it uses the exergonic flow of electrons from NADH and FADH2 to pump H+ from the matrix across the inner membrane and into the intermembrane space of the mitochondria creating an energy-storing proton gradient across the inner membrane that is then used to synthesize ATP.
-Also within the inner mitochondrial matrix is the enzyme ATP synthase
-ATP synthase provides the only channel for Hydrogen ions (protons) in the intermembrane space of the mitochondria to diffuse down their concentration gradient back into the mitochondrial matrix.
-ATP synthase uses the energy stored in the proton gradient across the inner membrane (also called a proton-motive force) to synthesize ATP from ADP and inorganic phosphate (Pi)
-The H+ diffusion is an exergonic process because it is proceeding in a favorable direction, toward equilibrium (equal concentration on both sides of the membrane)
-Chemiosmosis: the process of using a hydrogen ion gradient to perform cellular work
-ATP synthase is a multisubunit complex with four parts, each consisting of multiple polypeptides
-the hydrogen ions move one by one into binding sites on the rotor, causing it to spin, and producing ATP
- Although the ETC does not produce ATP on its own, it uses the exergonic flow of electrons from NADH and FADH2 to pump H+ from the matrix across the inner membrane and into the intermembrane space of the mitochondria creating an energy-storing proton gradient across the inner membrane that is then used to synthesize ATP.
-Also within the inner mitochondrial matrix is the enzyme ATP synthase
-ATP synthase provides the only channel for Hydrogen ions (protons) in the intermembrane space of the mitochondria to diffuse down their concentration gradient back into the mitochondrial matrix.
-ATP synthase uses the energy stored in the proton gradient across the inner membrane (also called a proton-motive force) to synthesize ATP from ADP and inorganic phosphate (Pi)
-The H+ diffusion is an exergonic process because it is proceeding in a favorable direction, toward equilibrium (equal concentration on both sides of the membrane)
-Chemiosmosis: the process of using a hydrogen ion gradient to perform cellular work
-ATP synthase is a multisubunit complex with four parts, each consisting of multiple polypeptides
-the hydrogen ions move one by one into binding sites on the rotor, causing it to spin, and producing ATP
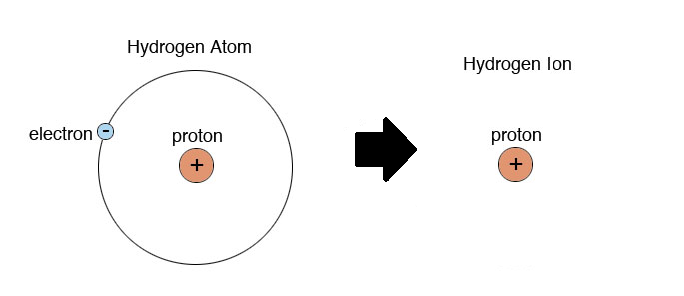
Why are hydrogen ions (H+) referred to as protons you ask?
-because a hydrogen atom consists of only one electron and one proton, so when the hydrogen atom gives up its electron it is essentially just a single proton.
-H+ denotes that the hydrogen has lost an electron.
-because a hydrogen atom consists of only one electron and one proton, so when the hydrogen atom gives up its electron it is essentially just a single proton.
-H+ denotes that the hydrogen has lost an electron.
An Accounting of ATP Production by Cellular Respiration
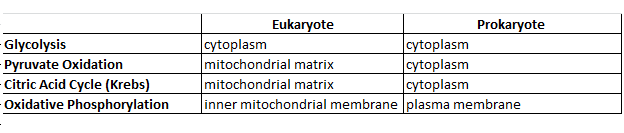
Summary of Process Locations in Eukaryotes and Prokaryotes:
Summary of Products:
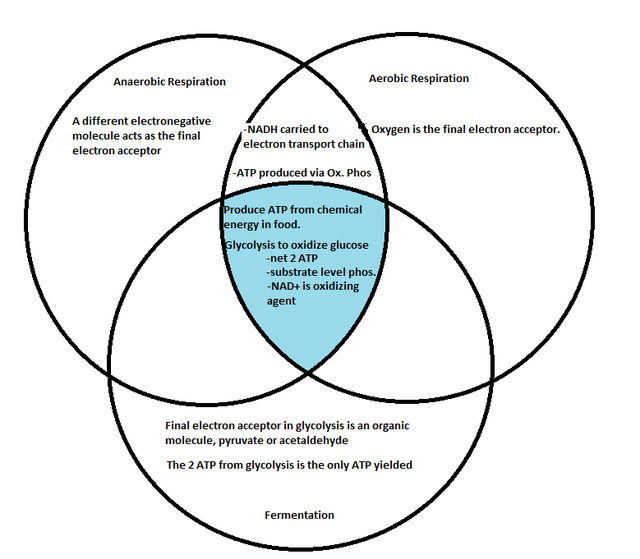
- 9.5: Fermentation and anaerobic respiration enable cells to produce ATP without the use of oxygen
1) Anaerobic respiration:
-prokaryotes that live in environments lacking O2
-still have electron transport chains, but use different final electron acceptors
2) Fermentation:
-no oxygen and no electron transport chain
-Fermentation is an extension of glycolysis
-NAD+ is recycled from NADH by transferring electrons to pyruvate product.
Types of Fermentation:
1) Alcohol fermentation: yeast (fungi), used in brewing and baking
-pyruvate converted to ethanol:
-pyruvate loses CO2 and becomes acetaldehyde
-acetaldehyde is reduced by NADH and becomes ethanol
2) Lactic acid fermentation: fungi/ bacteria, used to make dairy products
-pyruvate directly reduced by NADH to form lactate
-No CO2 production
-When oxygen is scarce human muscle cells will resort to lactic acid fermentation to generate ATP. (the build up of lactic acid due to change in process was previously though to cause the "burn" associated with strenuous exercise, but has since been challenged)
Comparing Fermentation with Anaerobic and Aerobic Respiration:
-Cellular respiration produces more ATP than does Fermentation.
-Obligate anaerobes: carry out only fermentation or anaerobic respiration, cannot survive with oxygen present
-Facultative anaerobes: can use fermentation or respiration depending on oxygen presence. (yeast, bacteria)
The Evolutionary Significance of Glycolysis
-Glycolysis plays a role in all processes, which makes it likely that glycolysis is the most deeply rooted of these processes in evolution.
-Ancient prokaryotes are thought to have used glycolysis exclusively to make ATP long before oxygen was present in Earth's atmosphere.
-The fact that glycolysis occurs in the cytoplasm also contributes to its early evolution, prior to eukaryotic cells and their membrane bound organelles.
- 9.6: Glycolysis and the citric acid cycle connect to may other metabolic pathways
-Although we follow a molecule of glucose, free glucose molecules are not common to our diets
-We obtain calories from fats, proteins, sucrose (and other disaccahrides) and starch (a polysaccharide).
-Proteins must first be broken down into amino acids.
-most are used to construct new proteins
-those that are available in excess will be converted to intermediates of glycolysis by enzymes.
- the first step in this process is deamination, the removal of the amino group.
-In animals, the nitrogenous waste is excreted in the urine.
-Fats are broken down into glycerol and fatty acids
-the glycerol can then be converted into an intermediate of glycolysis
- However, most of the energy stored in fats is found in the fatty acids
-fatty acids are broken down via beta oxidation
-beta oxidation breaks down fatty acids to 2 C segments
-these 2-C segments enter the citric acid cycle as acetyl coA
-A gram of fat produces the most amount of ATP
Biosynthesis (Anabolic Pathways)
-Not all food is harvested for its chemical energy to produce ATP.
-Some food is used as carbon skeletons for producing biological molecules.
-Intermediates of glycolysis and the citric acid cycle can be routed to anabolic pathways as precursors for cells to build molecules they need.
Regulation of Cellular Respiration via Feedback Mechanisms
-Supply and demand determine how food and intermediates along pathways will be routed.
-The cell will not expend unnecessary energy to synthesize a substance that is not needed.
- A common mechanism to enforce this control over energy expenditure is feedback inhibition
Feedback inhibition: when the end product of an anabolic pathway inhibits the enzyme that catalyzes an
early step in the pathway.
-The cell regulates catabolism, as well.
-If ATP concentration drops, respiration will speed up
-If ATP concentration meets the demand, respiration will slow down.
-Catabolisms is also controlled by regulating enzyme activity at strategic points in catabolic pathways.
-Phosphofructokinase is the enzyme that catalyzes step 3 of glycolysis, committing the substrate irreversibly to the glycolytic pathway.
-controlling this step can speed up or slow down the entire process of catabolism.
-phosphofructokinase is considered the "pace maker" of respiration
-It is inhibited by ATP and stimulated by AMP, it is also inhibited by citrate. If the citric acid cycle is way ahead, citrate travels to the cytoplasm to slwo down glycolysis via phosphofructokinase.