CHAPTER 5: THE STRUCTURE AND FUNCTION OF LARGE BIOLOGICAL MOLECULES
carbohydrates
lipids
proteins
nucleic acids
- macromolecule: extremely large molecules, include proteins, nucleic acids, and carbohydrates
- structure determines function in all biological molecules
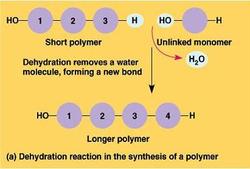
- Monomer: smaller molecules that serve as the building blocks or repeating units that make up a polymer.
-Alone, monomers have chemical and physical properties that differ from that of the polymer
they form together.
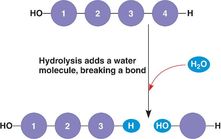
-Monomers are joined covalently by dehydration reactions and separated by hydrolysis
-These processes are facilitated by enzymes, specialized macromolecules that speed up the reaction process
-Cells have thousand of different macromolecules that vary based on cell type.
-Key to the function of the macromolecules is the arrangement of the 40-50 common monomers.
- Dehydration reaction: the removal of a molecule of water (-OH from one molecule, and -H from the other) to covalently bond two monomers in the construction of a polymer
- The Molecules of Life
carbohydrates
lipids
proteins
nucleic acids
- macromolecule: extremely large molecules, include proteins, nucleic acids, and carbohydrates
- structure determines function in all biological molecules
- 5.1: Macromolecules are polymers, built from monomers
- Monomer: smaller molecules that serve as the building blocks or repeating units that make up a polymer.
-Alone, monomers have chemical and physical properties that differ from that of the polymer
they form together.
-Monomers are joined covalently by dehydration reactions and separated by hydrolysis
-These processes are facilitated by enzymes, specialized macromolecules that speed up the reaction process
-Cells have thousand of different macromolecules that vary based on cell type.
-Key to the function of the macromolecules is the arrangement of the 40-50 common monomers.
- Dehydration reaction: the removal of a molecule of water (-OH from one molecule, and -H from the other) to covalently bond two monomers in the construction of a polymer
- Hydrolysis: the process of adding a molecule of water (-OH to one molecule, and -H to the other) to break the bond between two monomers in a polymer.
-Hydrolysis releases monomers into the bloodstream for distribution to other cells that then use
dehydration reactions to assemble new, different polymers needed to perform the functions of that cell.
-Hydrolysis releases monomers into the bloodstream for distribution to other cells that then use
dehydration reactions to assemble new, different polymers needed to perform the functions of that cell.
- 5.2: Carbohydrates serve as fuel and building material
- Monosaccharide: Simple sugars, these serve as the monomers (building blocks) of carbohydrates.
-The of these monosaccharides follows the molecular ratio CH2O
- Monosaccharides, particularly glucose, are major nutrients for cells. Cellular respiration is the
multi-reaction process in which cells break down glucose for usable energy.
-Those that are not broken down for energy, are broken down for use of their carbon-skeletons
in the synthesis of other small molecules (such as fatty acids and amino acids)
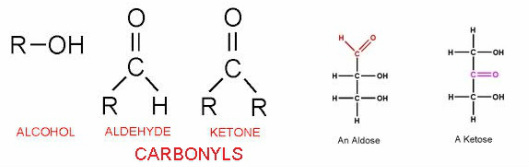
-Important functional groups of these molecules are:
- hydroxyl groups (-OH), and carbonyl groups (CO)
-The location of the carbonyl determines whether
the sugar is a ketose (carbonyl at C-2) or an aldose (carbonyl at C-1)
-Monosaccharides are grouped based on the number of carbons present in their carbon skeletons (3= triose, 5=pentose, 6=hexose)
- The arrangement of molecules and atoms around the assymetric carbon/s (carbon atom attached to 4 atoms or
groups of atoms) give the sugars distinct binding properties, despite appearing to be small differences.
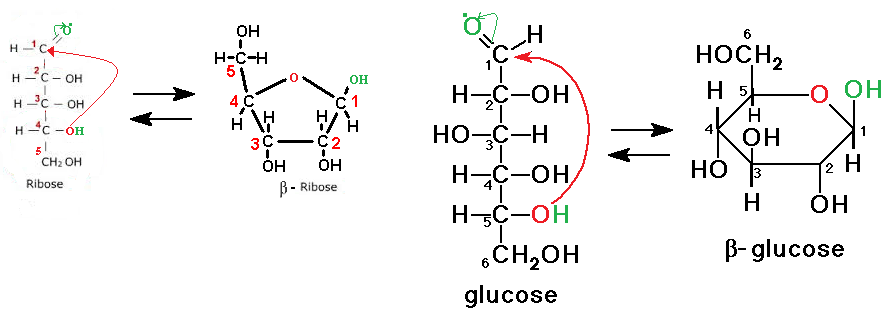
- Most 5 and 6 carbon sugars form ring structures in aqueous solutions. See images below for ring formation.
-There are two different ring structures for each sugar, alpha and beta.
-Whether or not a structure is alpha or beta is dependent on the location of the
hydroxyl group on C-1.
If the hydroxyl group is below the plane, it is alpha.
If the hydroxyl group is above the plane, it is beta.
- The arrangement of molecules and atoms around the assymetric carbon/s (carbon atom attached to 4 atoms or
groups of atoms) give the sugars distinct binding properties, despite appearing to be small differences.
- Most 5 and 6 carbon sugars form ring structures in aqueous solutions. See images below for ring formation.
-There are two different ring structures for each sugar, alpha and beta.
-Whether or not a structure is alpha or beta is dependent on the location of the
hydroxyl group on C-1.
If the hydroxyl group is below the plane, it is alpha.
If the hydroxyl group is above the plane, it is beta.
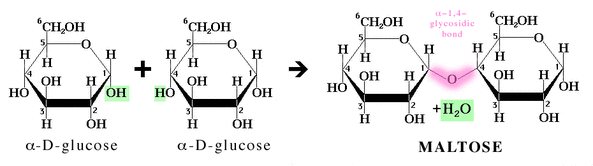
- Disaccharide: two monosaccharides joined covalently by a dehydration reaction, to form a glycosidic bond
- Glycosidic bonds are formally named according to the number of the two carbons involved in the
reaction and the location of the hydroxyl groups on the two anomeric carbons (C-1's) of the
monosaccharides.
1-4 glycosidic linkage is demonstrated below
The linkage is then named alpha- 1, 4 or beta- 1, 4
-Common disaccharides:
Glucose + Glucose = Maltose (malt sugar)
Glucose + Galactose = Lactose (sugar in milk)
Glucose + Fructose = Sucrose (table sugar)
- Glycosidic bonds are formally named according to the number of the two carbons involved in the
reaction and the location of the hydroxyl groups on the two anomeric carbons (C-1's) of the
monosaccharides.
1-4 glycosidic linkage is demonstrated below
The linkage is then named alpha- 1, 4 or beta- 1, 4
-Common disaccharides:
Glucose + Glucose = Maltose (malt sugar)
Glucose + Galactose = Lactose (sugar in milk)
Glucose + Fructose = Sucrose (table sugar)
- Polysaccharide: Carbohydrate polymer, macromolecule, consisting of many (100 to >1000) monosaccharides, formed by individual dehydration reactions for the addition of each monosaccharide
-Split into two functional categories composed of Glucose: Storage and Structural
-Storage Polysaccharides
Starch: stored in plant cells as granules within their plastids as a stock-pile
of glucose, hydrolyzed and used for energy as needed (helical shape)
- Amylose: form of unbranched starch ( alpha- 1, 4 linkage)
-Amylopectin: form of branched starch ( alpha- 1, 4 linkage, alpha- 1, 6 linkage at branch points)
- Humans have enzymes that hydrolyze plant starch, making
it a source of glucose for cells
- Potato tubers and grains represent the main sources of
starch in our diets
Glycogen: excessively branched glucose polymer stored in human liver and muscle cells, hydrolyzed
and used for energy as needed ( alpha- 1, 4 linkage, alpha- 1, 6 linkage at branch points)
-Ineffective for long term energy source, as it is quickly depleted.
-Structural Polysaccharides
Cellulose: unbranched polymer, present in the cell walls of plants, component of paper and cotton
( straight shape, beta- 1, 4 linkage, making every glucose monomer upside-down with respect to its neighbor)
- Rigid structure due to hydrogen bonding between -OH groups on paralleling strands
-Multiple strands form bundles called microfibrils
- Humans unable to hydrolyze beta- 1, 4 linkage, and cannot
digest cellulose, "insoluble fiber"
- Instead, cellulose lubricates our digestive tracts by encouraging
mucus secretion due to its rigid structure abrading the lining
- Certain animals, like cows, have protists or prokaryotes capable of
hydrolyzing these bonds for them in their digestive tracts, this
allows them to utilize cellulose's nutrients.
Chitin: component of arthropods' exoskeletons and the cell walls of fungi, contains a nitrogenous appendage,
called an N-acetyl group, on each glucose monomer ( beta- 1,4 linkage)
-Split into two functional categories composed of Glucose: Storage and Structural
-Storage Polysaccharides
Starch: stored in plant cells as granules within their plastids as a stock-pile
of glucose, hydrolyzed and used for energy as needed (helical shape)
- Amylose: form of unbranched starch ( alpha- 1, 4 linkage)
-Amylopectin: form of branched starch ( alpha- 1, 4 linkage, alpha- 1, 6 linkage at branch points)
- Humans have enzymes that hydrolyze plant starch, making
it a source of glucose for cells
- Potato tubers and grains represent the main sources of
starch in our diets
Glycogen: excessively branched glucose polymer stored in human liver and muscle cells, hydrolyzed
and used for energy as needed ( alpha- 1, 4 linkage, alpha- 1, 6 linkage at branch points)
-Ineffective for long term energy source, as it is quickly depleted.
-Structural Polysaccharides
Cellulose: unbranched polymer, present in the cell walls of plants, component of paper and cotton
( straight shape, beta- 1, 4 linkage, making every glucose monomer upside-down with respect to its neighbor)
- Rigid structure due to hydrogen bonding between -OH groups on paralleling strands
-Multiple strands form bundles called microfibrils
- Humans unable to hydrolyze beta- 1, 4 linkage, and cannot
digest cellulose, "insoluble fiber"
- Instead, cellulose lubricates our digestive tracts by encouraging
mucus secretion due to its rigid structure abrading the lining
- Certain animals, like cows, have protists or prokaryotes capable of
hydrolyzing these bonds for them in their digestive tracts, this
allows them to utilize cellulose's nutrients.
Chitin: component of arthropods' exoskeletons and the cell walls of fungi, contains a nitrogenous appendage,
called an N-acetyl group, on each glucose monomer ( beta- 1,4 linkage)
- 5.3: Lipids are a diverse group of hydrophobic molecules
-hydrophobic property arises from their structures, consisting of mostly hydrocarbons
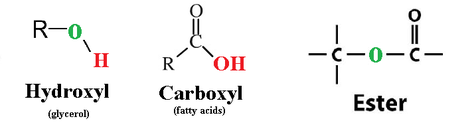
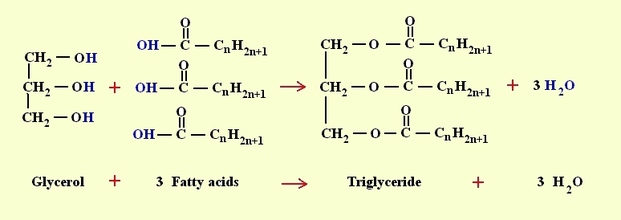
-Fats (triglyceride/triacylglycerol): made up of a molecule of glycerol (bearing 3 -OH groups), and 3 fatty acid chains (composed of long hydrocarbon chains with a carboxyl group on one end).
-Hydrocarbon chains of the fatty acids are what make fats hydrophobic, the water
molecules H-bond one another, excluding the fats, and separating them from solution
-the glycerol is linked to the fatty acids by separate dehydration reactions, where a
hydroxyl group of glycerol donates -H, and the fatty acid donates an -OH from its
carboxylcontaining end.
-the bonds formed are called ester linkage
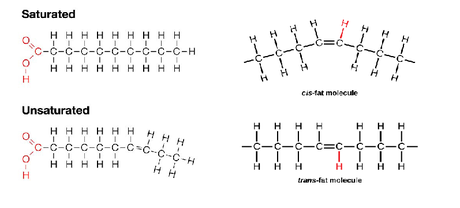
-Saturated vs. Unsaturated fats:
-A fat is considered saturated when its fatty acids are at capacity for hydrogen atoms.
- Storage: animal fats are saturated, flexibility of hydrocarbon chains
allow for compact storage in our adipose cells that can be tapped
into as an energy source that is much more sustainable than glycogen.
- Protection: adipose tissue also serves to cushion vital organs and maintain
body heat
-A fat is considered unsaturated when instead of the fatty acids being saturated with
hydrogen atoms, one or more double bonds exist between the carbons in the skeleton.
-Naturally, the double bonds are in cis formation, meaning that the hydrogens
remaining on the double bonded carbons are on the same side of the molecule,
and a kink is formed in the chain at the site of the double bond
-unsaturated fats can be converted to saturated fats synthetically by hydrogenation,
which breaks the double bond and adds H2.
-Diets high in saturated fats can lead to atherosclerosis, or the formation of plaque on artery
walls, increasing the risk of cardiovascular disease
-However synthetic trans fats of processed foods (unsaturated fats withdouble bonds in
trans formation, where the remaining H atoms are on separate sides of the molecule,
are indicated in contributing the most to atherosclerosis
-A fat is considered saturated when its fatty acids are at capacity for hydrogen atoms.
- Storage: animal fats are saturated, flexibility of hydrocarbon chains
allow for compact storage in our adipose cells that can be tapped
into as an energy source that is much more sustainable than glycogen.
- Protection: adipose tissue also serves to cushion vital organs and maintain
body heat
-A fat is considered unsaturated when instead of the fatty acids being saturated with
hydrogen atoms, one or more double bonds exist between the carbons in the skeleton.
-Naturally, the double bonds are in cis formation, meaning that the hydrogens
remaining on the double bonded carbons are on the same side of the molecule,
and a kink is formed in the chain at the site of the double bond
-unsaturated fats can be converted to saturated fats synthetically by hydrogenation,
which breaks the double bond and adds H2.
-Diets high in saturated fats can lead to atherosclerosis, or the formation of plaque on artery
walls, increasing the risk of cardiovascular disease
-However synthetic trans fats of processed foods (unsaturated fats withdouble bonds in
trans formation, where the remaining H atoms are on separate sides of the molecule,
are indicated in contributing the most to atherosclerosis
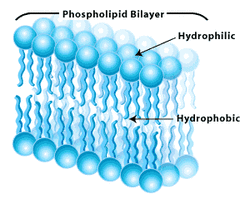
-Phospholipids: major component of cell membranes (phospholipid bilayer), glycerol molecule attached to two fatty acids and one phosphate group, the phosphate group carries a negative charge in the cell (typically another small charged molecule is attached to the phosphate group).
-The two ends of phospholipids have different properties:
- Phosphate group: negatively charged, polar, hydrophilic
- Hydrocarbon tails: neutral in charge, non polar, hydrophobic
- Because of these different properties, phospholipids form bilayers in water, where
Their hydrophobic tails are inward (away from the water) and their hydrophilic heads
are outward (in contact with water)
-this is the arrangement of phospholipids in cell membranes,
allowing for the existence of cells and making these molecules
very important biologically!
-The two ends of phospholipids have different properties:
- Phosphate group: negatively charged, polar, hydrophilic
- Hydrocarbon tails: neutral in charge, non polar, hydrophobic
- Because of these different properties, phospholipids form bilayers in water, where
Their hydrophobic tails are inward (away from the water) and their hydrophilic heads
are outward (in contact with water)
-this is the arrangement of phospholipids in cell membranes,
allowing for the existence of cells and making these molecules
very important biologically!
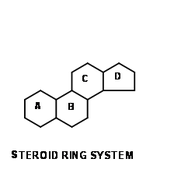
-Steroids: lipids with structures consisting of 4 fused carbon rings, they are identified based on the groups that are attached to these rings.
- Cholesterol: component of animal cell membranes and precursor for other steroids.
-synthesized in the liver and obtained from diet
-high levels contribute to the development of atherosclerosis
-levels controlled by presence of saturated and trans fats (raise levels)
and unsaturated fats (lower levels)
- Cholesterol: component of animal cell membranes and precursor for other steroids.
-synthesized in the liver and obtained from diet
-high levels contribute to the development of atherosclerosis
-levels controlled by presence of saturated and trans fats (raise levels)
and unsaturated fats (lower levels)
- 5.4 Proteins include a diversity of structures, resulting in a wide range of functions
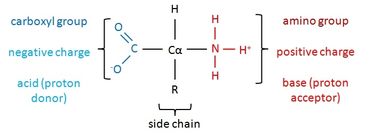
-Protein: a biologically functional molecule made up of one or more polypeptides, each folded into a specific 3-D structure.
- Amino Acid: building blocks of polypeptides, organic molecules with both an amino group and a carboxyl group.
- There are 20 different amino acids, but all amino acids share a common structure:
-assymetric carbon at the center, called the alpha carbon
-an amino group (NH3+ in cell, see image)
-a carboxyl group (COO- in the cell, see image)
-a hydrogen atom
- an "R" group, or side chain
- Side chains determine the type of amino acid
and are different for each
-Therefore, the side chain also determines the
chemical and physical properties of an amino acid
-Side chains are categorized as
1) polar (hydrophilic) : -OH groups, -SH groups, or NH2-C=O groups at the tips of side chains
2) nonpolar (hydrophobic): Hydrocarbons, just a bunch of CH arrangements at the tips
3) charged (hydrophilic): COO- or NH3+ at the tips of the side chains
-Charged side chains are Acidic - (COO-, proton donors) ,
or Basic + (NH3+, proton acceptors)
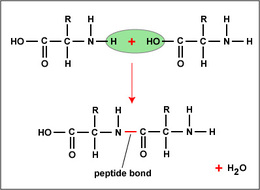
-Polypeptide: polymers of amino acids, joined covalently by peptide bonds formed by individual dehydration reactions, where the -OH of a carboxyl group and an -H from an amino group are removed as H2O.
- The repeating sequence, due to the common parts of amino acid structure, along a polypeptide
are called the polypeptide backbone.
- The side chains that extend from this backbone still account for the molecule's chemical properties
-The end of the polypeptide with the amino group is the N-terminus, and the other end
with the carboxyl group is called the C-terminus
- Amino Acid: building blocks of polypeptides, organic molecules with both an amino group and a carboxyl group.
- There are 20 different amino acids, but all amino acids share a common structure:
-assymetric carbon at the center, called the alpha carbon
-an amino group (NH3+ in cell, see image)
-a carboxyl group (COO- in the cell, see image)
-a hydrogen atom
- an "R" group, or side chain
- Side chains determine the type of amino acid
and are different for each
-Therefore, the side chain also determines the
chemical and physical properties of an amino acid
-Side chains are categorized as
1) polar (hydrophilic) : -OH groups, -SH groups, or NH2-C=O groups at the tips of side chains
2) nonpolar (hydrophobic): Hydrocarbons, just a bunch of CH arrangements at the tips
3) charged (hydrophilic): COO- or NH3+ at the tips of the side chains
-Charged side chains are Acidic - (COO-, proton donors) ,
or Basic + (NH3+, proton acceptors)
-Polypeptide: polymers of amino acids, joined covalently by peptide bonds formed by individual dehydration reactions, where the -OH of a carboxyl group and an -H from an amino group are removed as H2O.
- The repeating sequence, due to the common parts of amino acid structure, along a polypeptide
are called the polypeptide backbone.
- The side chains that extend from this backbone still account for the molecule's chemical properties
-The end of the polypeptide with the amino group is the N-terminus, and the other end
with the carboxyl group is called the C-terminus
-Structures and Functions of Proteins:
- A functional protein is not just a polypeptide chain, but one or more polypeptides precisely twisted, folded and coiled a molecule with a unique shape that makes it functional, or able to recognize and bind a specific molecule or set of molecules.
-This folding is the product of interactions and bonds between parts of the polypeptide chain, determined by
its sequence of amino acids.
- The final shape of a protein can be considered either globular (spherical) or fibrous (long strands).
-Four levels of protein structure:
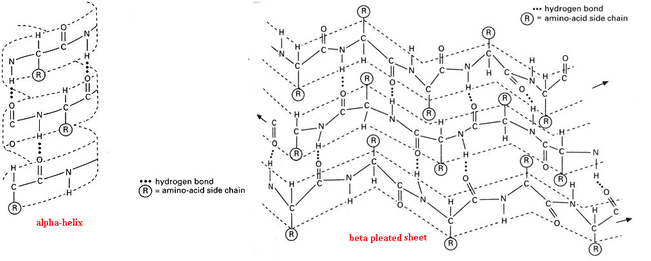
-Primary Structure: the linear amino acid sequence of a polypeptide.
-Secondary Structure: resulting coils and folds that are products of hydrogen bonding between polar backbone
constituents, the oxygens (C=O) have partially negative charges and the hydrogens (-NH) have partially positive charges.
-Alpha Helix: segment coiling created by H-bonds between every 4th amino acid in the polypeptide
chain
-Beta Pleated Sheet: H-bonding between two parallel segments of polypeptide chain
- A functional protein is not just a polypeptide chain, but one or more polypeptides precisely twisted, folded and coiled a molecule with a unique shape that makes it functional, or able to recognize and bind a specific molecule or set of molecules.
-This folding is the product of interactions and bonds between parts of the polypeptide chain, determined by
its sequence of amino acids.
- The final shape of a protein can be considered either globular (spherical) or fibrous (long strands).
-Four levels of protein structure:
-Primary Structure: the linear amino acid sequence of a polypeptide.
-Secondary Structure: resulting coils and folds that are products of hydrogen bonding between polar backbone
constituents, the oxygens (C=O) have partially negative charges and the hydrogens (-NH) have partially positive charges.
-Alpha Helix: segment coiling created by H-bonds between every 4th amino acid in the polypeptide
chain
-Beta Pleated Sheet: H-bonding between two parallel segments of polypeptide chain
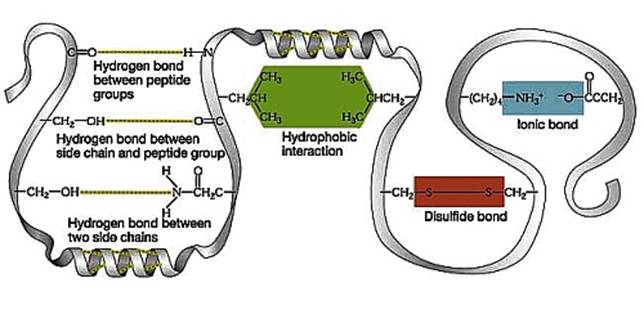
-Tertiary Structure:the structure resulting from side chain interactions.
-Hydrophobic interactions: the hydrophobic/nonpolar side chains tend to aggregate inside the folded
structure away from potential interactions with the aqueous environment.
-van der Waals interactions: when nonpolar molecules are close together the constant
movement of their electrons allow for momentary assymetry in electron distribution, these
moments that result in these attractions. Once grouped, these individually weak attractive
forces hold the nonpolar side chains together
-Hydrogen bonding and Ionic bonds: represent polar and charged side chain interactions.
-Disulfide Bridges: covalent bonding between close cysteine monomers, their sulfhydryl groups
(-SH) lose their hydrogens and bond to eachother (S-S)
-Hydrophobic interactions: the hydrophobic/nonpolar side chains tend to aggregate inside the folded
structure away from potential interactions with the aqueous environment.
-van der Waals interactions: when nonpolar molecules are close together the constant
movement of their electrons allow for momentary assymetry in electron distribution, these
moments that result in these attractions. Once grouped, these individually weak attractive
forces hold the nonpolar side chains together
-Hydrogen bonding and Ionic bonds: represent polar and charged side chain interactions.
-Disulfide Bridges: covalent bonding between close cysteine monomers, their sulfhydryl groups
(-SH) lose their hydrogens and bond to eachother (S-S)
-Quaternary Structure: overall protein structure that results from the association of 2 or more polypeptides.
-Chaperonins (chaperone proteins): these are proteins that aid in the proper folding of other proteins.
- They keep the polypeptide safe while it goes through the processes above by shielding it from
harsh cellular conditions in the cytoplasm in a capped tube-like complex.
-Denaturation and Misfolding:
-Denaturation is the unfolding of a protein due to the destruction of the weak forces holding it together
-Causes: alterations in the pH, salt concentration, or temperature within the cell,
chemical exposure, a nonpolar solvent
-Effects: a biologically inactive protein
-In some cases the protein can regain its shape and become functional again,
this is called renaturation.
-Misfolded proteins are implicated in many diseases: cystic fibrosis, Alzheimer's, Parkinson's, sickle cell anemia
-Functions of Proteins: proteins like enzymes selectively speed up chemical reactions, proteins like antibodies protect us from disease, transport proteins help transport molecules across the cell membrane, receptor proteins help convey chemical messages, structural proteins provide the framework of connective tissues, contractile and motor proteins permit movement of cilia and muscles, storage proteins store amino acids, hormonal proteins signal regulation functions of other cells.
-Chaperonins (chaperone proteins): these are proteins that aid in the proper folding of other proteins.
- They keep the polypeptide safe while it goes through the processes above by shielding it from
harsh cellular conditions in the cytoplasm in a capped tube-like complex.
-Denaturation and Misfolding:
-Denaturation is the unfolding of a protein due to the destruction of the weak forces holding it together
-Causes: alterations in the pH, salt concentration, or temperature within the cell,
chemical exposure, a nonpolar solvent
-Effects: a biologically inactive protein
-In some cases the protein can regain its shape and become functional again,
this is called renaturation.
-Misfolded proteins are implicated in many diseases: cystic fibrosis, Alzheimer's, Parkinson's, sickle cell anemia
-Functions of Proteins: proteins like enzymes selectively speed up chemical reactions, proteins like antibodies protect us from disease, transport proteins help transport molecules across the cell membrane, receptor proteins help convey chemical messages, structural proteins provide the framework of connective tissues, contractile and motor proteins permit movement of cilia and muscles, storage proteins store amino acids, hormonal proteins signal regulation functions of other cells.
- 5.5: Nucleic acids store, transmit, and help express hereditary information
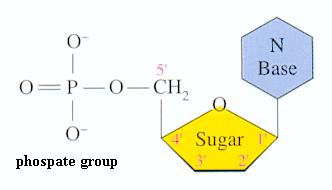
-Nucleotide: building blocks of nucleic acids
-Nucleotides are made up of 3 parts:
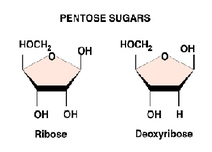
1) a 5- Carbon sugar, called a pentose
-ribose: RNA
-deoxyribose: DNA (lacks oxygen atom on C-2)
2) a nitrogenous base (bc nitrogens tend to take up H+ from solution, proton acceptors)
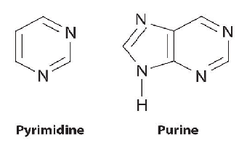
- purines: two ring nitrogen-containing structure
-adenine and guanine
- pyramidines: a single nitrogen-containing ring structure
- cytosine, thymine (DNA only) and uracil (RNA only)
-purines hydrogen bond with pyramidines in base pairing
-DNA: A-T and C-G
-RNA: A-U and C-G
3) a phosphate group
-attached to C-5 of the pentose
-A nucleoside is just the pentose and nitrogenous base without a phosphate group
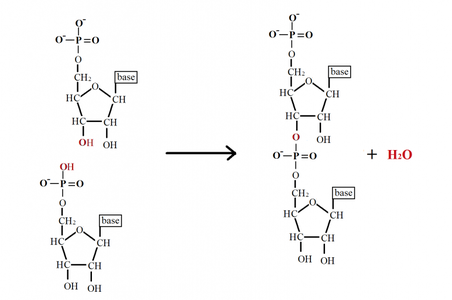
-Polynucleotide: polymer of nucleotides that are connect by dehydration reactions
-Phosphodiester linkage: phophosphate groups form phosphodiester bonds to connect two pentoses of
nucleotides. (the one it was connected to at C-5 and now C-3 on the other nucleotide's pentose)
-This bonding results in a repeating sugar-phosphate backbone in nucleic acids
-The end of the nucleic acid with a phosphate group on C-5 is referred to as the 5' (5 prime) end
-The end of the nucleic acid with a hydroxyl group on C-3 is reffered to as the 3' (3 prime) end
- The nitrogenous bases that protrude from this back bone are in a specific sequential order, this specific
sequence is what makes up our genetic code, and specifies the amino acid sequences of proteins
synthesized in our cells
-Phosphodiester linkage: phophosphate groups form phosphodiester bonds to connect two pentoses of
nucleotides. (the one it was connected to at C-5 and now C-3 on the other nucleotide's pentose)
-This bonding results in a repeating sugar-phosphate backbone in nucleic acids
-The end of the nucleic acid with a phosphate group on C-5 is referred to as the 5' (5 prime) end
-The end of the nucleic acid with a hydroxyl group on C-3 is reffered to as the 3' (3 prime) end
- The nitrogenous bases that protrude from this back bone are in a specific sequential order, this specific
sequence is what makes up our genetic code, and specifies the amino acid sequences of proteins
synthesized in our cells
-DNA (Deoxyribonucleic Acid): Nucleic acid consisting of two polynucleotides (strands), that form what we know as the double helix, contain genetic information
- The two strands run antiparallel to each other, meaning in opposite 5'--->3' directions
- The two strands are also complementary, referring to the specific pairing of bases A-T and G-C
- The sugar-phosphate backbones of the strands form the outside of the helix, while base pairs
hydrogen bond to their counterparts on the interior, holding the molecule together
- The double stranded, antiparallel, and complementary structure allows DNA to complete two identical
copies of itself simultaneously during replication.
-RNA (Ribonucleic Acid): single-stranded nucleic acid
-Complementary base pairing sometimes occurs between two RNA molecules, or even an RNA molecule
with itself, to conform to their functional 3-D structures.
- In RNA, A binds U and G binds C
-Functions:
-DNA contains instructions for all cellular activities within discrete units of inheritance, called genes.
-DNA controls its own replication making identical copies of genetic information to pass to new cells during cell division
-Gene Expression: DNA directs RNA synthesis, and controls protein synthesis through RNA
-DNA contains genes the code for specific mRNA
-mRNA leaves the nucleus and interacts with the ribosome in the cytoplasm to synthesize the polypeptide
-tRNA brings amino acids according to mRNA sequence to construct polypeptide.
-The polypeptide eventually becomes a protein that implements genetic programs.
- The two strands run antiparallel to each other, meaning in opposite 5'--->3' directions
- The two strands are also complementary, referring to the specific pairing of bases A-T and G-C
- The sugar-phosphate backbones of the strands form the outside of the helix, while base pairs
hydrogen bond to their counterparts on the interior, holding the molecule together
- The double stranded, antiparallel, and complementary structure allows DNA to complete two identical
copies of itself simultaneously during replication.
-RNA (Ribonucleic Acid): single-stranded nucleic acid
-Complementary base pairing sometimes occurs between two RNA molecules, or even an RNA molecule
with itself, to conform to their functional 3-D structures.
- In RNA, A binds U and G binds C
-Functions:
-DNA contains instructions for all cellular activities within discrete units of inheritance, called genes.
-DNA controls its own replication making identical copies of genetic information to pass to new cells during cell division
-Gene Expression: DNA directs RNA synthesis, and controls protein synthesis through RNA
-DNA contains genes the code for specific mRNA
-mRNA leaves the nucleus and interacts with the ribosome in the cytoplasm to synthesize the polypeptide
-tRNA brings amino acids according to mRNA sequence to construct polypeptide.
-The polypeptide eventually becomes a protein that implements genetic programs.